Separating 400 g (~3/4 cup) of NaCl ion pairs from an initial separation distance of 2.8...
Fantastic news! We've Found the answer you've been seeking!
Question:
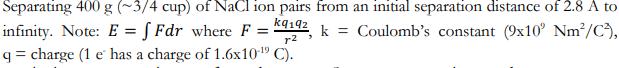
Transcribed Image Text:
Separating 400 g (~3/4 cup) of NaCl ion pairs from an initial separation distance of 2.8 A to Coulomb's constant (9x10' Nm²/C3), infinity. Note: E = f Fdr where F = kq192k r2 q=charge (1 e has a charge of 1.6x10-¹9 C). Separating 400 g (~3/4 cup) of NaCl ion pairs from an initial separation distance of 2.8 A to Coulomb's constant (9x10' Nm²/C3), infinity. Note: E = f Fdr where F = kq192k r2 q=charge (1 e has a charge of 1.6x10-¹9 C).
Expert Answer:
Answer rating: 100% (QA)
The amount of energy required to separate 400 g of NaCl ion pairs from an initial separation distanc... View the full answer

Related Book For 

Accounting Information Systems
ISBN: 978-0133428537
13th edition
Authors: Marshall B. Romney, Paul J. Steinbart
Posted Date:
Students also viewed these chemistry questions
-
Customers arrive at a checkout counter in a department store according to a Poisson distribution at an average of seven per hour. During a given hour, what are the probabilities that a no more than...
-
The corner convenience store (CCS) gets its cool drinks delivered by a wholesaler at CCS, light cola sells at a rate of 40 six-packs per day. The shop is open 250 days per year. a six pack of light...
-
The process tank is used to store liquids during manufacturing. Estimate both the volume of the tank and its surface area. The tank has a flat top and a thinwall. m- -3 m 6 m 4 m
-
1). Cody Jenkins and Lacey Tanner formed a partnership to provide landscaping services. Jenkins and Tanner shared profits and losses equally. After all the tangible assets have been adjusted to...
-
The caloric contents and the sugar contents (in grams) of 11 high-fiber breakfast cereals (a) x = 150 calories (b) x = 90 calories (c) x = 175 calories (d) x = 208 calories Find the equation of the...
-
The adjusted trial balance for Fraser Valley Services Ltd. is given in E4-8. In E4-8 The unadjusted and adjusted trial balances follow for Fraser Valley Services Ltd. at the end of its fiscal year,...
-
The following table gives the characteristics of a building in Houston, TX. Determine the cooling load for July 30 at solar noon. Any information regarding the load not given may be assumed or...
-
The income statement, balance sheets, and additional information for Great Adventures, Inc., are provided below. Additional Information for 2017: 1. Borrowed $500,000 in January 2017. Made 12 monthly...
-
On Jan, 1, 20X1 ABC Corp sold equipment with a cost of P20,000,000 and accumulated depreciation of P7,000,000 in exchange for cash of P500,000 and non-interest bearing note receivable of P8,000,000...
-
LOT is a partnership owned by Long, Oliver, and Taker. The partners' profit-and loss- sharing ratio is 2:2:1, respectively. The adjusted trial balance of the partnership at November 30, 2018,...
-
I. For this exercise, use the following modular arithmetic hashing function to find the index of the element in the hash table: Hash(x) = x mod 13. a. Start with an empty hashing table, using Open...
-
Vaughn, Inc., manufactures clamps used in the overhead bin latches of several leading airplane models. Thomas Taylor, president of Vaughn, Inc., has gathered the following cost information from the...
-
The economy will go into decline if taxes are too high. taxes must be too high. the economy is in decline since people are being laid off and the gross domestic product is going down. identify the...
-
A bicycle plant must produce 1952 bicycles per month to meet demand. The set-up costs for each production run are $1220 , and it costs $20 to store a bicycle for a month. How many production runs...
-
These are selected account balances on December 31, 2025. Land $147000 Land (held for future use) 220500 Buildings 1176000 Inventory 294000 Equipment 661500 Furniture 147000 Accumulated Depreciation...
-
Autonomous aggregate expenditures decreases by $200 million, the marginal propensity to consume is 0.50, marginal propensity to invest is 0.25, and the marginal propensity to import is 0.10....
-
A firm invested $300,000 in an old factory and started producing secret products. After the top management realized that it costs $1,000 to produce one item, the decision was made to abandon the...
-
Imagine a sound wave with a frequency of 1.10 kHz propagating with a speed of 330 m/s. Determine the phase difference in radians between any two points on the wave separated by 10.0 cm.
-
You are performing a financial audit of the general ledger accounts of Preston Manufacturing. As transactions are processed summary journal entries are added to the general ledger file at the end of...
-
a. Download the spreadsheet for problem 14.7 from the website for this textbook. b. Create formulas to calculate Accumulated depreciation (all assets use the straight line method; all assets...
-
A large organization had 18 months to replace its old customer information system with a new one that could differentiate among customer levels and provide appropriate products and services on...
-
Derive the expression for the complex frequency response of an undamped torsional system.
-
A single-story building frame is subjected to a harmonic ground acceleration, as shown in Fig. 3.57. Find the steady-state motion of the floor (mass \(m\) ). Figure 3.57:- 22 -x(t) m (!) - A cos cot...
-
Find the horizontal displacement of the floor (mass \(m\) ) of the building frame shown in Fig. 3.57 when the ground acceleration is given by \(\ddot{x}_{g}=100 \sin \omega t \mathrm{~mm} /...

Study smarter with the SolutionInn App


