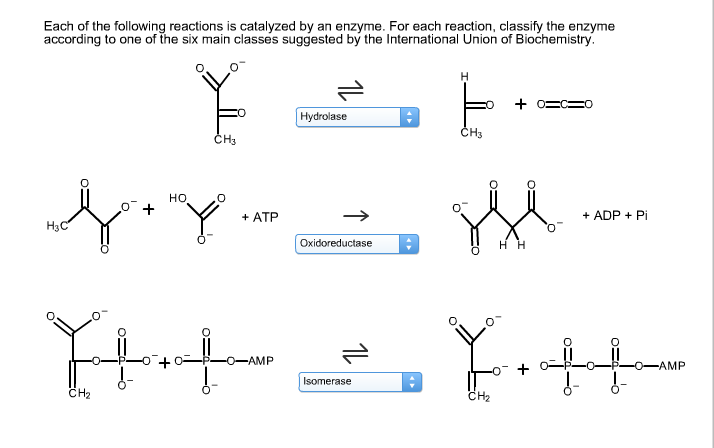
Each of the following reactions is catalyzed by an enzyme. For each reaction, classify the enzyme...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Each of the following reactions is catalyzed by an enzyme. For each reaction, classify the enzyme according to one of the six main classes suggested by the International Union of Biochemistry. H3C CH₂ HO CH3 0 + ATP parafor O-AMP Hydrolase Oxidoreductase 11 Isomerase H CH3 +03 CH₂ HH + ADP + Pi Y.tt + O-AMP Each of the following reactions is catalyzed by an enzyme. For each reaction, classify the enzyme according to one of the six main classes suggested by the International Union of Biochemistry. H3C CH₂ HO CH3 0 + ATP parafor O-AMP Hydrolase Oxidoreductase 11 Isomerase H CH3 +03 CH₂ HH + ADP + Pi Y.tt + O-AMP
Expert Answer:
Answer rating: 100% (QA)
Lyases These enzymes cleave CC CO CN and CS bonds by mechanisms other ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) CH3C = PCH +...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Each of the following reactions has been carried out under conditions such that disubstitution or trisubstitution occurred. Identify the principal organic product in each case. (a) Nitration of...
-
The stockholders' equity accounts of Whispering Company have the following balances on December 31, 2025. Common stock, $10 par, 304,000 shares issued and outstanding $3,040,000 Paid-in capital in...
-
The December 31, 2015, unadjusted trial balance for Demon Deacons Corporation is presented below. At year-end, the following additional information is available: a. The balance of Prepaid Rent,...
-
Meadowlands Design produces head covers for golf clubs. The company expects to generate a profit next year. It anticipates fixed manufacturing costs of $200,500 and fixed general and administrative...
-
The production department of Zunni's Manufacturing is considering two numerically controlled drill presses; one must be selected. Comparison data is shown in the table below. MARR is 10 percent/year....
-
On December 31, 2012, the American Bank enters into a debt restructuring agreement with Barkley Company, which is now experiencing financial trouble. The bank agrees to restructure a 12%, issued at...
-
With what group of others do you most closely identify? Those from your home town? Those of your same sex? Religion? Race? Income level? IQ level? Who are the people with whom you feel most relaxed?...
-
Assume today is t=0. A 10-year fixed rate bond with a 5% coupon rate is selling at par (annual coupons). From $200 FV of this bond, we form a floater and an inverse floater by equally splitting its...
-
A compound contains 24.27% of Carbon, 4.07% hydrogen, and 71.65% of chlorine. Its molar is 98.96 g (C= 12.011g, H=1.008g, Cl= 35.453 g) How many moles of Hydrogen are there in the compound?
-
Windows are a major source of heat loss from a house because glass has a higher thermal conductivity than the other materials typically used in construction and because insulation can be placed...
-
A shop owner raises the price of a $120 pair of shoes by 30%. After a few weeks, because of falling sales, the owner reduces the price of the shoes by 30%. A customer then says that the shoes are...
-
A product s unit cost includes $ 5 0 for direct materials used, $ 4 0 for direct labor incurred, $ 3 0 for manufacturing overhead applied, and its unit selling price is $ 3 0 0 . Depreciation is $ 1...
-
W hat is the total asset turn over and return on equity for a company with an ROA of 1 3 % profit margin of 4 % and a debt ratio of 4 5 % ?
-
A $ 1 3 , 1 7 6 par - value bond is purchased at a coupon rate of 5 % . If interest payments are to be made semiannually ( every six months ) , how much will each interest payment be ? Round your...
-
Suppose you are given the following quotes by a dealer: Brazilian Real/U.S. Dollar = 3.914 U.S. Dollar/Australian Dollar = 0.735 Chinese Yuan/U.S. Dollar = 7.594 You plan to make a 15-day trip to...
-
Use nodal analysis to determine voltages v1, v2, and v3 in the circuit Fig. 3.76. Figure 3.76 4 S 3i, 2 A 4A
-
What is a particle accelerator, and how does one operate? Why are they required for certain nuclear reactions?
-
Predict the sign of S, if possible, for each of the following reactions. If you cannot predict the sign for any reaction, state why. a. C2H2(g) + 2H2(g) C2H6(g) b. N2(g) + O2(g) 2NO(g) c. 2C2H2(g)...
-
The Sn 2+ (aq) ion can be written in more detail as Sn(H 2 O) 6 2+ . This ion is acidic by hydrolysis. Write a possible equation for this hydrolysis.
-
Depository institutions have been losing their advantage over other financial intermediaries in attracting customers funds. Why?
-
In the aftermath of the financial crisis of 20072009, there were calls to reinstate the separation of commercial and investment banking activities that was removed with the repeal of the...
-
The globalization of banking has led to the need for global benchmarks for interest rates. In light of the LIBOR scandal, what characteristic do you think is most central to any new interest rate...

Study smarter with the SolutionInn App


