Effect of [ATP]/[ADP] Ratio on Free Energy of Hydrolysis of ATP Using Equation 13-4, plot AG...
Fantastic news! We've Found the answer you've been seeking!
Question:
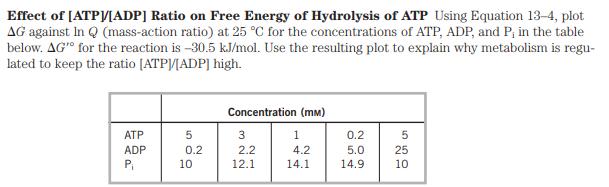
Transcribed Image Text:
Effect of [ATP]/[ADP] Ratio on Free Energy of Hydrolysis of ATP Using Equation 13-4, plot AG against In Q (mass-action ratio) at 25 C for the concentrations of ATP, ADP, and P, in the table below. AG" for the reaction is -30.5 kJ/mol. Use the resulting plot to explain why metabolism is regu- lated to keep the ratio [ATP]/[ADP] high. ATP ADP P 5 0.2 10 Concentration (mm) 1 4.2 14.1 3 2.2 12.1 0.2 5.0 14.9 5 25 10 Effect of [ATP]/[ADP] Ratio on Free Energy of Hydrolysis of ATP Using Equation 13-4, plot AG against In Q (mass-action ratio) at 25 C for the concentrations of ATP, ADP, and P, in the table below. AG" for the reaction is -30.5 kJ/mol. Use the resulting plot to explain why metabolism is regu- lated to keep the ratio [ATP]/[ADP] high. ATP ADP P 5 0.2 10 Concentration (mm) 1 4.2 14.1 3 2.2 12.1 0.2 5.0 14.9 5 25 10
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these biology questions
-
Calculate the standard free-energy change at 25C for the following reaction. 3Cu(s) + 2NO3 (aq) + 8H+(aq) 3Cu2+(aq) + 2NO(g) + 4H2O(l)
-
Calculate the standard free-energy change at 25C for the following reaction. 4Al(s) + 3O2(g) + 12H + (aq) 4Al3 + (aq) + 6H2O(l) Use standard electrode potentials.
-
a. Calculate K1 at 25C for phosphoric acid: b. Which thermodynamic factor is the most significant in accounting for the fact that phosphoric acid is a weak acid? Why? H3PO4(aq)--H"(aq) + H,PO4-(aq)...
-
Inverse permutation. Write a program that reads in a permutation of the integers 0 to n-1 from n command-line arguments and prints the inverse permutation. (If the permutation is in an array a[], its...
-
Evaluate the Tsaos financial strategy. Could they have done anything more effectively?
-
For the year 2 0 2 3 , a company had the following: Sales $ 1 , 4 5 4 , 0 0 0 COGS 8 0 0 , 0 0 0 If we assume sales will increase by 8 % for 2 0 2 4 , what would be the projected dollar amount for...
-
Air enters a 4-cm-square galvanized steel duct with \(p_{0}=\) \(150 \mathrm{kPa}, T_{0}=400 \mathrm{~K}\), and \(V_{1}=120 \mathrm{~m} / \mathrm{s}\). \(\mu=2.2 \times 10^{-5}\) \(\mathrm{N} \cdot...
-
ProBuilder has the following June 30, 2016, fiscal-year-end unadjusted balances: Allowance for Sales Discounts, $0; and Accounts Receivable, $10,000. Of the $10,000 of receivables, $2,000 are within...
-
Describe the steps for using the strategy of Vocabulary Development with the student in physical education (make sure to describe the steps fully)
-
Griffin and Lasky, Inc. (G&L), supplies industrial automation equipment and machine tools to the automotive industry. G&L recognizes revenue on its long-term contracts over time. Customer orders have...
-
TERMINOLOGY: Match the word in Column A with its brief definition in Column B. Write the letter of the definition on the line to the left of the word. USE CAPITAL LETTERS ONLY !! Points will be lost...
-
1) Create tables of the cash flows for the bonds. Find the prices of the bonds. O 2) Find the yields on the bonds using XIRR and YIELD. Bond A Bond face value Coupon rate Purchase date 1,000.00 11%...
-
1. Draw five aggregate supply and demand graphs at long run equilibrium. Illustrate what would happen to the price level, output and employment under the following circumstances: 1)Congress raises...
-
To find the blue shaded area above, we would calculate: b I's a f(x)dx = area Where: a f(x) = area , b: -
-
Marvelous Makeovers (MM) is a thriving upscale decor business, located in a neighbourhood known for its: rich history; numerous public events; vibrant downtown district; charming bed and breakfast...
-
Are there issues associated with keeping the large sum of money in accumulated earnings? The corporation has a large amount of money in retained earnings. The corporations retained earnings...
-
Annie Magnolia gave 200 shares of stock in her closely held corporation to a local not-for-profit organization (NFP) with a cost to her of $250,000 and a fair market value on the date of the gift of...
-
H.J. Heinzs annual dividends were as follows: 1990 ..............$0.540 1991.............. 0.620 1992 .............. 0.700 1993.............. 0.780 1994 .............. 0.860 1995 .............. 0.940...
-
In a series of oil-drop experiments, the charges measured on the oil drops were 3.20 1019 C, 6.40 1019 C, 9.60 1019 C, and 1.12 1018 C. What is the smallest difference in charge between any two...
-
A solution contains 4.25 g of ammonia per 250.0 mL of solution. Electrical conductivity measurements at 25C show that 0.42% of the ammonia has reacted with water. Write the equation for this reaction...
-
What is the cell potential (Ecell) of a spontaneous cell that is run at 25oC and contains [Cr3+] = 0.10 M and [Ag+] = 1.0 104 M?
-
In Fig. 1130, there is no explicit invariant that states that a Transaction cannot be linked to movements that are linked to the same account, because the structure of the model already demands is....
-
Apply the Account/Transaction pattern to the parallel financial transactions of the example shown in Figs. 1131 to 1135. As the bills may be paid forward, it may be necessary to use memo entries....
-
Look at Fig. 1119 and create a new discount strategy that gives a percentage of discount for sales with more than a certain number of products. Order | < > < > +nr: Natural = < > +date: Date...

Study smarter with the SolutionInn App



