Energy (kJ/mol) 150 AE 100 50 0 E Reaction A What is the change in energy...
Fantastic news! We've Found the answer you've been seeking!
Question:
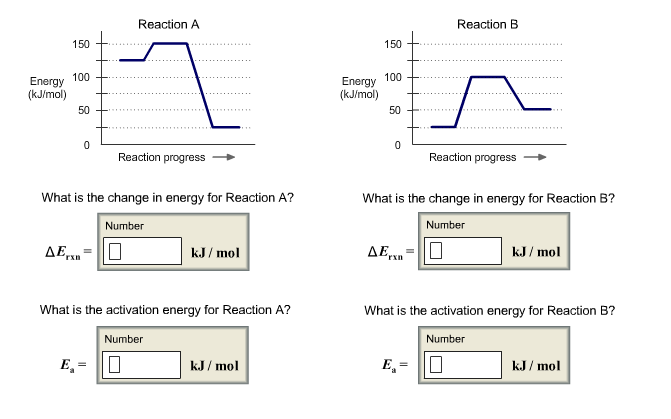
Transcribed Image Text:
Energy (kJ/mol) 150 AE 100 50 0 E Reaction A What is the change in energy for Reaction A? Number Reaction progress kJ/mol What is the activation energy for Reaction A? Number 0 kJ/mol Energy (kJ/mol) 150 100 50 0 DE What is the change in energy for Reaction B? Number 0 Reaction B = E₂² Reaction progress kJ/mol What is the activation energy for Reaction B? Number kJ/mol Energy (kJ/mol) 150 AE 100 50 0 E Reaction A What is the change in energy for Reaction A? Number Reaction progress kJ/mol What is the activation energy for Reaction A? Number 0 kJ/mol Energy (kJ/mol) 150 100 50 0 DE What is the change in energy for Reaction B? Number 0 Reaction B = E₂² Reaction progress kJ/mol What is the activation energy for Reaction B? Number kJ/mol
Expert Answer:
Answer rating: 100% (QA)
A chemical reaction needs energy to start which is called activation energy This ... View the full answer

Related Book For 

Modeling the Dynamics of Life Calculus and Probability for Life Scientists
ISBN: 978-0840064189
3rd edition
Authors: Frederick R. Adler
Posted Date:
Students also viewed these chemistry questions
-
What is the change in entropy of 0.130 kg of helium gas at the normal boiling point of helium when it all condenses isothermally to 1.00 L of liquid helium? (Hint: See Table 17.4 in Section 17.6.)
-
What is the change in entropy of 1.00m3 of water at 0oC when it is frozen to ice at 0oC?
-
What is the change in entropy of mercury vapor (Lv = 2.7 x 105 J/kg) when 0.50 kg of it condenses to a liquid at its boiling point of 357 oC?
-
Research about the competitive and comparative advantage of the Argentina in terms of physical and human resources and how it is making use of these resources for international trade purpose. 3....
-
Match each of the following preferred stock features with its description. Preferred Stock Features _________ 1. Convertible _________ 2. Redeemable _________ 3. Cumulative Description a. Prior...
-
Atlas Pumps Corporation, a manufacturer of industrial pumps, reports the following results for the year ended January 31, 2014: Retained earnings, February 1, 2013 . . . . . . . . . . . . . . . . . ....
-
In 2004, Jui-Chen Lin, a citizen of China, entered into an agreement with Robert Chiu and Charles Cobb, citizens of the United States, to form an LLC to acquire and operate a fast-food restaurant in...
-
(Terminology, Relationships, Computations, Entries) Complete the following statements by filling in the blanks. (a) In a period in which a taxable temporary difference reverses, the reversal will...
-
Explain the ways in which the firms and industries that led cities' recoveries from de-industrialization are similar and different from the firms and industries that led the industrial revolution....
-
In spring 2021, Amir Nathoo was at the helm of one of the most talked-about startups in the massive and fragmented educational technology (EdTech) industry. Just six years earlier, Nathoo and his...
-
A 36.00 kg lead sphere is hanging from a hook by a thin wire 2.80 m long and is free to swing in a complete circle. Suddenly it is struck horizontally by a 5.00 kg steel dart that embeds itself in...
-
What is the straight-line method of depreciation, and how is it different from other depreciation methods? 2. What is the formula used to calculate depreciation expense under the straight-line...
-
A business retains 79% of its customers from one year to the next. If the business uses a 11% discount rate, calculate the margin multiple. Rounding: 2 decimal places.
-
All types of projects operate on a budget. Project managers need to see what the overall cost of the project is going to be from the start to the scheduled launch date. Cost Baseline is a way for PMs...
-
How do postcolonial critiques of globalization challenge dominant narratives of progress and development, foregrounding the legacies of colonialism, imperialism, and uneven power relations in shaping...
-
ACCT 326 Accounting System Review This project requires you to evaluate a PC-based accounting system to see how the system handles recording transactions, collecting data, and generating reports and...
-
James has $3,000 in credit card debt, which charges 14% interest. How long will it take to pay off the card if he makes the minimum payment of $60 a month?
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
A population starts at size 100, and receives 10 immigrants in the first year, loses 5 emigrants in the second year, receives 10 immigrants in the third year, and so forth. In each of the above...
-
Suppose that fraction used = 0.5 / 1.0 + 0.1Mt. Write the discrete-time dynamical system and solve for the equilibrium and compare with M* = 2.0 for the basic model. The model describing the dynamics...
-
Without setting r and h to particular values, find the equilibrium algebraically. Does the equilibrium get larger when h gets larger? Does it get larger when r gets larger? If the answers seem odd...
-
An April 2014 study by researchers at University College London found that people who ate seven or more portions of fruits and vegetables a day had a \(42 \%\) lower risk of death at any point in...
-
The consumption of alcohol is illegal for Afghan citizens, but the government does allow the sale of alcohol under licence to foreigners. Use a diagram to show how such a prohibition would affect the...
-
Suppose that the demand curve for wheat is \(Q=100-10 p\) and the supply curve is \(Q=10 p\). The government imposes a specific tax of \(t=1\) per unit. a. How do the equilibrium price and quantity...

Study smarter with the SolutionInn App


