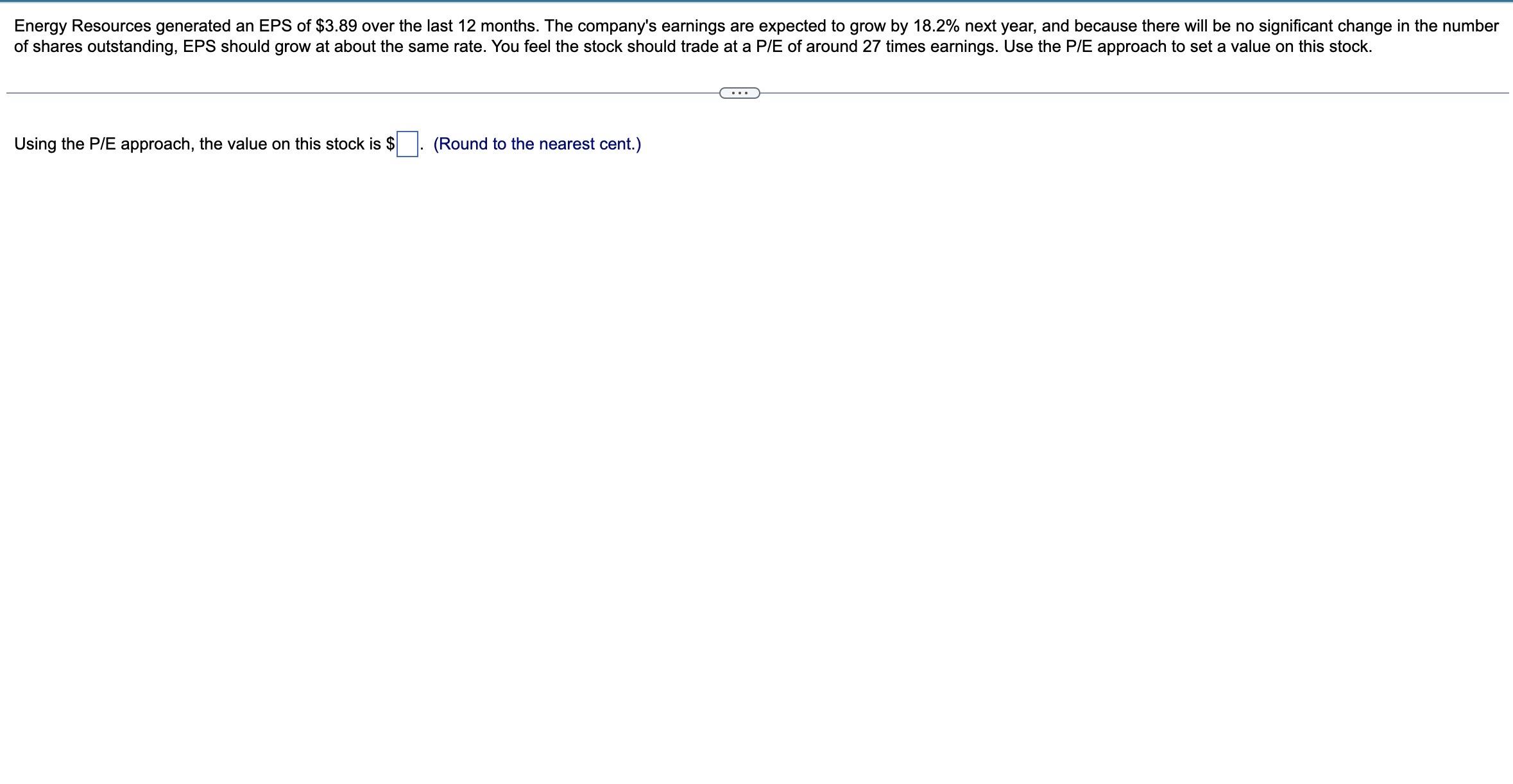
Energy Resources generated an EPS of $3.89 over the last 12 months. The company's earnings are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Energy Resources generated an EPS of $3.89 over the last 12 months. The company's earnings are expected to grow by 18.2% next year, and because there will be no significant change in the number of shares outstanding, EPS should grow at about the same rate. You feel the stock should trade at a P/E of around 27 times earnings. Use the P/E approach to set a value on this stock. Using the P/E approach, the value on this stock is $ (Round to the nearest cent.) Energy Resources generated an EPS of $3.89 over the last 12 months. The company's earnings are expected to grow by 18.2% next year, and because there will be no significant change in the number of shares outstanding, EPS should grow at about the same rate. You feel the stock should trade at a P/E of around 27 times earnings. Use the P/E approach to set a value on this stock. Using the P/E approach, the value on this stock is $ (Round to the nearest cent.)
Expert Answer:
Answer rating: 100% (QA)
To calculate the value of the stock using the PE approach we need to m... View the full answer

Related Book For 

Fundamentals Of Investing
ISBN: 9780135175217
14th Edition
Authors: Scott B. Smart, Lawrence J. Gitman, Michael D. Joehnk
Posted Date:
Students also viewed these finance questions
-
AviBank Plastics generated an EPS of $2.75 over the last 12 months. The companys earnings are expected to grow by 25% next year, and because there will be no significant change in the number of...
-
Lynx Auto generated an EPS of $3.50 over the last 12 months. The companys earnings are expected to grow by 20% next year, and, because there will be no significant change in the number of shares...
-
Stores (Wal-Mart) is the largest retailing firm in the world. Building on a base of discount stores, Wal-Mart has expanded into warehouse clubs and Supercenters, which sell traditional discount store...
-
Factor each polynomial. 64y 9 + z 6
-
If the outer electron in sodium moves in the n = 3 Bohr orbit, the effective nuclear charge would be Ze = 1e, and the energy of the electron would be 13.6 eV/3 2 = 1.51 eV. However, the ionization...
-
A small business owner who is concerned about passing the business on to heirs after retirement or death should prepare a formal ____________ plan. (a) retirement (b) succession (c) franchising (d)...
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
Doug's Custom Construction Company is considering three new projects, each requiring an equipment investment of $22,000. Each project will last for 3 years and produce the following net annual cash...
-
4. Consider a quantum system Q described by a Hilbert space H. (a) Suppose we are given a subspace Ho of H and a linear map from kets in Ho to others in h. That is, Vo)), in a linear way. This map...
-
Forecasted financial statements are given Sales (all on credit) Cost of Goods Sold Gross Profit Selling and Administrative Expense Rent Expense Depreciation Expense Operating profit (EBIT) Interest...
-
Asagai (Pty) Ltd pays a maintenance contractor to maintain its premises. The maintenance contractor is paid on an annual basis. On 1 November 2022, the company paid the maintenance contractor R40 000...
-
Let f(x) = x-4x+1 and g(x)=2x-1. Find each of the following. a) f(-3) b) g (0) c) 2f(x) - g(x) d) g(2k-1)
-
If f(x)=(x+3)+4, find when f(x) = 12 graphically and algebraically. (15 points for graphing correctly and identify the solution, 15 points for solving algebraically correctly) -14-12-10 14- 10- 2 x...
-
An aluminium object with a mass of 2.13 kg and at a temperature of 29.8 C comes to thermal contact with a 7.65 kg copper object which is initially at a temperature of 99.1 C. What is going to be the...
-
An average compact car carries 10 US gallon of gasoline in its fuel tank. The density of gasoline at 20 C is 0.77 g/mL, and its lower heating value is 44, 000 kJ/kg (that is, 44, 000 kJ of energy is...
-
(c) The graph shows the relationship between the peak wavelength of light emitted by a star and the surface temperature of the star. 1000 Peak wavelength in nm 800 600- 400 200- 0 5 10 15 Surface...
-
For this week's lab, you will use two of the classes in the Java Collection Framework: HashSet and TreeSet. You will use these classes to implement a spell checker. Set Methods this lab, you will...
-
In Exercises discuss the continuity of each function. f(x) -3 1 x - 4 y 3 2 -1 -2 -3+ 3 X
-
Lachlan purchased a 5%, zero-coupon bond with a 10-year maturity and A$10,000 par value 10 years ago. The bond matures tomorrow. How much will Lachlan receive in total from the investment, assuming...
-
Referring to Problem 5.20, assume you have a portfolio with $20,000 invested in each of investments A, B, and C. What is your portfolio beta? Data From Problem 5.20 Assume the betas for securities A,...
-
Briefly discuss several aspects of an industry that are important to its behavior and operating characteristics. How economic issues fit into industry analysis.
-
What is the half-life of uranium-238?
-
Who discovered that energy and mass are two different forms of the same thing?
-
Is it possible for a hydrogen nucleus to emit an alpha particle? Why? (a) yes, because alpha particles are the simplest form of radiation (b) no, because it would require the nuclear fission of...

Study smarter with the SolutionInn App


