Question: Esterification in a CSTR Consider the liquid-phase organic esterification reaction taking place in a CSTR depicted in Figure 4.40. Two streams, an acid stream containing
Esterification in a CSTR Consider the liquid-phase organic esterification reaction taking place in a CSTR depicted in Figure 4.40. Two streams, an acid stream containing no base, and a base stream containing no acid, are fed into the CSTR. The esterification reaction and its rate are given by
A + B → ester + H2O r-kcACB
in which A is the organic acid and B is the organic base. The acid and base are dissolved in an organic solvent and the acid and base feed streams have feed concentrations cAf and cBf, respectively. You may assume that the density of the fluid is independent of concentration over the concentration range of interest here.
The reactor's volume is constant during the entire operation.
(a) What are the units of k?
(b) What is the volumetric flowrate Q of the effluent stream in terms of the feed stream flowrates QA and QB? Show your reasoning
(c) Write out the transient material balances for components A and B. You should have differential equations for dcaldt, dcB/dt when you are finished. What initial conditions do you require for these two differential equations.
(d) Now consider the steady-state problem. Write the steady-state balances for the acid and base concentrations in the reactor, CAs, CBS.
(e) Can you solve these two equations for cAs, CBs in the general form for all values of the parameters? Is this steady-state solution unique?
(f) Whether or not you were able to solve the equations in the general form, given the following specific parameter values, find the steady-state reactor concentration:s CAs, CBs
τ=10 min QA = QB CAf = 8 mol/L cbf = 4 mol/L k -0.1 (in units of min, mol, L)
(g) What are the steady-state molar conversions of A and B for these specific param eter values?
Figure 4.40
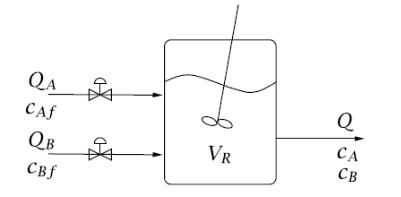
Figure 4.41

QA CAf CBF VR Q
Step by Step Solution
3.45 Rating (161 Votes )
There are 3 Steps involved in it
Lets address each part of the problem stepbystep a Units of k The reaction rate is given by r k cA cB Where r is the rate of reaction in moles per lit... View full answer

Get step-by-step solutions from verified subject matter experts


