Ethanol Production in Yeast When grown anaerobically on glucose, yeast (S. cerevisiae) converts pyruvate to acetaldehyde,...
Fantastic news! We've Found the answer you've been seeking!
Question:
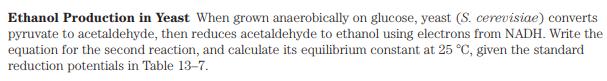
Transcribed Image Text:
Ethanol Production in Yeast When grown anaerobically on glucose, yeast (S. cerevisiae) converts pyruvate to acetaldehyde, then reduces acetaldehyde to ethanol using electrons from NADH. Write the equation for the second reaction, and calculate its equilibrium constant at 25 C, given the standard reduction potentials in Table 13-7. Ethanol Production in Yeast When grown anaerobically on glucose, yeast (S. cerevisiae) converts pyruvate to acetaldehyde, then reduces acetaldehyde to ethanol using electrons from NADH. Write the equation for the second reaction, and calculate its equilibrium constant at 25 C, given the standard reduction potentials in Table 13-7.
Expert Answer:
Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these biology questions
-
Write the equation for the reaction of 1-hexyne with ethylmagnesium bromide as if it involved ethyl anion (CH3CH2) instead of CH3CH2MgBr and use curved arrows to represent the flow of electrons.
-
Write the equation for the reaction of t-butyl alcohol with potassium metal. Name the product.
-
Write the equation for the reaction of 1-hexyne with ethylmagnesium bromide as if it involved ethyl anion instead of CH3CH2MgBr and use curved arrows to represent the flow of electrons. (CH3CH2-)
-
People who earn a higher salary can afford more goods, including health care. However, according to Grossman, they will choose a higher desired health stock. Why is this so, according to the model?
-
David Patton, a merchandising manager for J.C. Penney Company, was having an intimate relationship with a co-worker. The store manager, McKay, told Patton that if he did not cease this relationship...
-
A store sells yogurt in small, medium, and large sizes. A small yogurt costs $2, a medium costs $3, and a large costs $5. In one hour, the shop sold 27 yogurts for $98. There were five more large...
-
Determine the static pressure to stagnation pressure ratio associated with the following motion in standard air: (a) a runner moving at the rate of \(10 \mathrm{mph}\), (b) a cyclist moving at the...
-
The December 31, 2006 balance sheet of the Blackmon Corporation disclosed the following information relating to its receivables: *The company is contingently liable for a discounted note receivable...
-
What does the Performance Management (PM) process consist of? On what motivational theory is the PM process based on?
-
The beginning inventory at Midnight Supplies and data on purchases and sales for a three month period ending March 31 are as follows: Date Transaction Number of Units Per Unit Total Jan. 1 Inventory...
-
One month Tom rented 12 movies and 2 video games for a total of $29. The next month he rented 3 movies and 5 video games for a total of $32. Find the rental cost for each movie and each video game. 2...
-
Write equations that show how to use the distributive property ("each with each" or "FOIL") to find the product 3x2 4. Your equations need to: correctly demonstrate the distributive property or FOIL...
-
List name of payer. If any interest is from a seller-financed mortgage and the buyer used the property as a personal residence, see the instructions and list this interest first. Also, show that...
-
Consider the dynamics for the underlying asset: dS(t) = S(t)dt + v(t)S(t)dZ (t), dv(t) -Bv(t)dt + ddz(t), = where S(t) is the stock price, v(t) is the volatility, Z(t) and Z(t) are Wiener processes...
-
In the system of two blocks and a spring shown above, blocks 1 and 2 are connected by a string that passes over a pulley. The initially unstretched spring connects block 1 to a rigid wall. Block 1 is...
-
Plant 1 Plant 2 Plant 3 Plant 4 Plant 5 Demand (units/year) Plant 1 Plant 2 Plant 3 Plant 4 Plant 5 Excess Capaity Plant 1 Plant 2 Plant 3 Plant 4 Plant 5 Total Cost Market 1 Market 1 0 0 0 0 0...
-
The following information is available for Beta Enterprise: Cost of debt before tax 10.0% Cost of equity Market value of debt Market value of equity Tax rate 11.1% $3085770 $4848582 31% Compute the...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
You are a summer intern in a clinical hematology lab. The lab director gives you a sample of a patient's blood proteins and asks you to characterize the thrombin in the sample. She also tells you...
-
Write a balanced equation for the complete metabolic oxidation of each of the following. Include O2, ADP, and Pi as reactants and ATP, CO2, and H2O as products. (a) Stearic acid (b) Oleic acid (c)...
-
pppApCpCpUpApGpApU-OH a. Using the straight-chain sugar convention shown on page 77, write the structure of the DNA strand that encoded this short stretch of RNA. b. Using the simplest convention for...
-
In this chapter, we noted that the Hilbert space of the harmonic oscillator corresponds to all those states that can be accessed from the ground state through action by an anatic function of...
-
We had shown that any state \(|\psiangle\) on the Hilbert space of the harmonic oscillator is described by some analytic function \(f\left(\hat{a}^{\dagger} ight)\) of the raising operator acting on...
-
We happen to find that the ground state of the harmonic oscillator (and the coherent states in general) is a minimum uncertainty state, but can we do the converse? That is, can we directly determine...

Study smarter with the SolutionInn App



