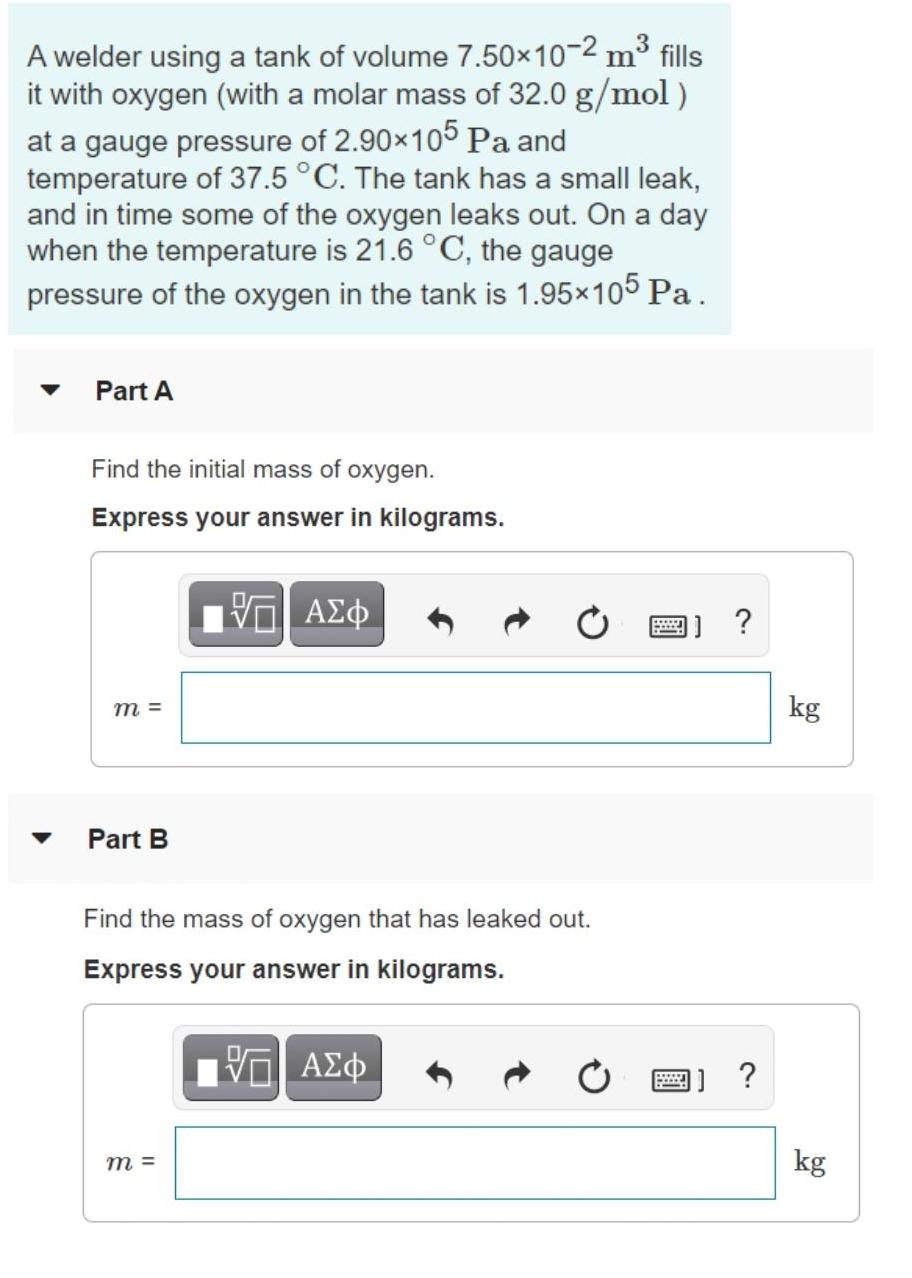
A welder using a tank of volume 7.50x10-2 m fills it with oxygen (with a molar...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A welder using a tank of volume 7.50x10-2 m³ fills it with oxygen (with a molar mass of 32.0 g/mol) at a gauge pressure of 2.90×105 Pa and temperature of 37.5 °C. The tank has a small leak, and in time some of the oxygen leaks out. On a day when the temperature is 21.6 °C, the gauge pressure of the oxygen in the tank is 1.95x105 Pa . Part A Find the initial mass of oxygen. Express your answer in kilograms. m = kg Part B Find the mass of oxygen that has leaked out. Express your answer in kilograms. ΑΣφ 画] ? m = kg A welder using a tank of volume 7.50x10-2 m³ fills it with oxygen (with a molar mass of 32.0 g/mol) at a gauge pressure of 2.90×105 Pa and temperature of 37.5 °C. The tank has a small leak, and in time some of the oxygen leaks out. On a day when the temperature is 21.6 °C, the gauge pressure of the oxygen in the tank is 1.95x105 Pa . Part A Find the initial mass of oxygen. Express your answer in kilograms. m = kg Part B Find the mass of oxygen that has leaked out. Express your answer in kilograms. ΑΣφ 画] ? m = kg
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these physics questions
-
A welder using a tank of volume 0.0750 m' fills it with oxygen (molar mass 32.0 g/mol) at a gauge pressure of 3.00 X 105 Pa and temperature of 37.0C. The tank has a small leak, and in time some of...
-
On a day when the wind is blowing toward the south at 3 m/s, a runner jogs west at 4 m/s. What is the velocity (speed and direction) of the air relative to the runner?
-
A rigid tank contains 1 kg oxygen at 160 K, 4 MPa. Determine the volume of the tank assuming we can use the Redlich-Kwong equation of state for oxygen. Compare the result with the ideal gas law.
-
Consider the pistoncylinder assembly containing a pure gas shown below. The initial volume of the gas is 0.05 m3, the initial pressure is 1 bar, and the cross-sectional area of the piston is 0.1 m2....
-
Determine the following items. a. SSTR b. MSTR c. SSE d. MSE e. F Sample 1 Sample 2 Sample 3 Sample 4
-
Critics have charged that compensation to top managers in the Units States is simply too high and should be cut back. For example, focusing on large corporations, Larry Ellison of Oracle has been one...
-
The Plant Assets account of Star Media shows the following: Star Media sold plant assets at a \(\$ 10,000\) loss. Where on the statement of cash flows should Star. Media report the sale of plant...
-
Denton Corporation employs 50 workers in its plant. Each employee is paid $10 per hour and works seven hours per day, Monday through Friday. Employees are paid every Friday. The last payday was...
-
1. Specific heat of Al is twice of that Fe. (a) If Al and Fe have same mass and the same amount of heat is added to Al and Fe individually, which one will end up having a higher temperature if...
-
On January 1. Ruiz Company issued bonds as follows: Face Value: Number of Years: Stated Interest Rate: Interest payments per year 500,000 15 7% Required: 1) Calculate the bond selling price given the...
-
On January 1, 2024, the general ledger of Dynamite Fireworks includes the following account balances: Accounts Cash Accounts Receivable Supplies Debit $ 23,800 5,200 Credit Land Accounts Payable...
-
The ancient Greek mathematician Euclid is credited with the development of the theorem that the sum of the angles of a triangle is 180 degrees. Use the previous information to solve for the measure...
-
You are running the bond trading desk of a large investment bank. This morning, you obtained the following quotes from your broker: There is a 0.5-year maturity zero-coupon bond with price 97.58...
-
2. Consider the Cagan model of nominal exchange rates. We will consider the impact on the exchange rate of an expectation of quantitative easing in the foreign economy. et = - (1) (177) [ms-oys + m...
-
Suppose the World includes two countries H and F, producing two goods 1 & 2, with the following information: (1). labor endowment: LH = 6, LF = 4 (1). labor requirement for producing 1 unit of good:...
-
3) How long did the arrow fly through the air before striking the ground? (When would h= 0 ft?) h=-32t+200t+5.75
-
Unit 3 Economics Performance Task: Writing Assignment for Business Cycle For Part II you will search for a news article (newspapers: New York Times, Washington Post, and Wall Street Journal are all...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Ethyl acetate, C4H8O2, is a fragrant substance used both as a solvent and as an aroma enhancer. Its Lewis structure is (a) What is the hybridization at each of the carbon atoms of the molecule? (b)...
-
For each element, indicate the number of valence electrons, core electrons, and unpaired electrons in the ground state: (a) Carbon, (b) Phosphorus, (c) Neon.
-
Automobile batteries contain sulfuric acid, which is commonly referred to as "battery acid." Calculate the number of grams of sulfuric acid in 1.00 gallon of battery acid if the solution has a...
-
Which of the following statements regarding the audit of negotiable notes receivable is not correct? a. Confirmation from the debtor is an acceptable alternative to inspection. b. Physical inspection...
-
To conceal defalcations involving receivables, the auditor would expect an experienced bookkeeper to charge which of the following accounts? a. Sales returns. b. Miscellaneous income. c. Petty cash....
-
An auditor's risk of misstatement of accounts receivable is least likely to increase if there is a. An increase in customer complaints about their accounts. b. A general slowdown in cash collections....

Study smarter with the SolutionInn App


