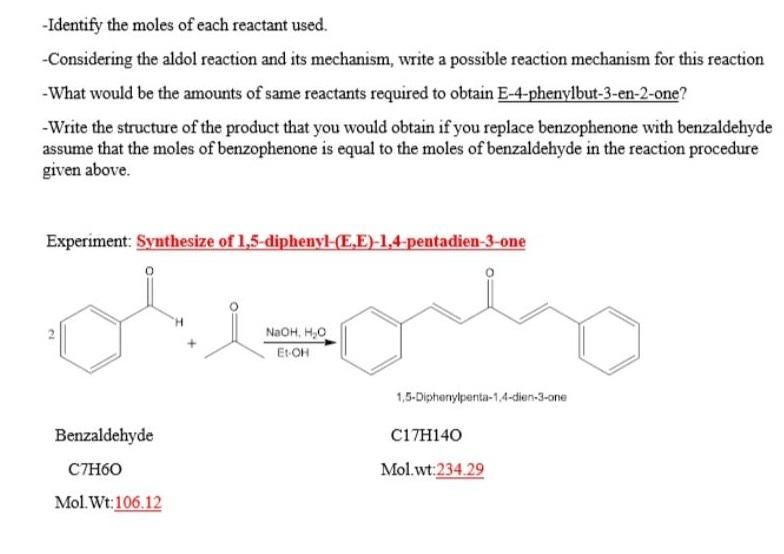
-Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
-Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a possible reaction mechanism for this reaction -What would be the amounts of same reactants required to obtain E-4-phenylbut-3-en-2-one? -Write the structure of the product that you would obtain if you replace benzophenone with benzaldehyde assume that the moles of benzophenone is equal to the moles of benzaldehyde in the reaction procedure given above. Experiment: Synthesize of 1,5-diphenyl-(E,E)-1,4-pentadien-3-one NaOH, H,0 Et-OH 1,5-Diphenylpenta-1,4-dien-3-one Benzaldehyde C17H140 CTH60 Mol.wt:234.29 Mol. Wt:106.12 2. -Identify the moles of each reactant used. -Considering the aldol reaction and its mechanism, write a possible reaction mechanism for this reaction -What would be the amounts of same reactants required to obtain E-4-phenylbut-3-en-2-one? -Write the structure of the product that you would obtain if you replace benzophenone with benzaldehyde assume that the moles of benzophenone is equal to the moles of benzaldehyde in the reaction procedure given above. Experiment: Synthesize of 1,5-diphenyl-(E,E)-1,4-pentadien-3-one NaOH, H,0 Et-OH 1,5-Diphenylpenta-1,4-dien-3-one Benzaldehyde C17H140 CTH60 Mol.wt:234.29 Mol. Wt:106.12 2.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In the diet example in this chapter, what would be the effect on the optimal solution of increasing the minimum calorie requirement for the breakfast to 500 calories? to 600 calories? Increase the...
-
1). A firm has entered into a 4-year, annual-pay, 6% plain vanilla interest rate swap with a notional principal value of $10,000,000. The firm is the fixed rate payer (i.e. the swap dealer is the...
-
A possible mechanism for a gas-phase reaction is given below. What is the rate law predicted by this mechanism? (fast equilibrium) 2NOCI NOC NO2NOCI (slow)
-
Yamini and Fatima, two students of Class IX of a school, together contributed Rs. 100 towards the Prime Ministers Relief Fund to help the earthquake victims. Write a linear equation which satisfies...
-
Air (79 percent N2 and 21 percent O2) is heated to 2000 K at a constant pressure of 2 atm. Assuming the equilibrium mixture consists of N2, O2, and NO, determine the equilibrium composition at this...
-
List the activities that must be completed before beginning this phase.
-
Outline the general strategy used in metagenomics.
-
Selected transactions for Babcock Company during November of the current year are listed in Problem 5-3A. In Problem 5-3A, the following were selected from among the transactions completed by Babcock...
-
An 76 kg man starts from rest and after 1.60 seconds is running at a speed of 11.0 m/s. The force needed for this man to get up to this speed in 1.60 seconds is ____ N.
-
An important part of planning manufacturing capacity is having a good forecast of sales. Elizabeth Burke is interested in forecasting sales of mowers and tractors in each marketing region as well as...
-
Carmen was in a department meeting. While her supervisor was talking, Carmen was checking her emails on her cell phone and only half-listening. When her supervisor asked her a question, Carmen was...
-
"What are semaphores, and could you provide a brief overview of their significance in operating systems?"
-
In September 2019, the Rotaract Club of UST AMV COA conducted a raffle draw to raise funds for its community services. A UST accounting student from Bataan, Philippines, won a MAC Air laptop valued...
-
A firm currently has a debt-equity ratio of 1/2. The debt, which is virtually riskless, pays an interest rate of 7 %. The expected rate of return on the equity is 14 %. What is the Weighted-Average...
-
The Zuri Co. needs to raise $66.6 million to finance its expansion into new markets. The company will sell new shares of equity via a general cash offering to raise the needed funds. The offer price...
-
Greg owes two debt payments a payment of $5072 that was due in 10 months ago and a payment of $1992 due in 8 months. If Greg makes a payment now, what would this payment be if money is worth 6.46%...
-
The following codes may contain an error or multiple errors. Find the error in each of the following code segment and rewrite a correct code using C language. Proof that the corrected codes provided...
-
After graduating from college and working a few years at a small technology firm. Preet scored a high-level job in the logistics department at Amex Corporation. Amex sells high-quality electronic...
-
A California grower has a 50-acre farm on which to plant strawberries and tomatoes. The grower has available 300 hours of labor per week and 800 tons of fertilizer, and he has contracted for shipping...
-
In Problem 15, Computers Unlimited wants to better meet demand at the four universities it supplies. It is considering two alternatives: (1) Expand its warehouse at Richmond to a capacity of 600, at...
-
In Problem 53, the decision analysis is for automobiles and light trucks. Blue Ridge Power and Light would like to reformulate the problem for its heavy construction equipment. Emergency maintenance...
-
Consider a second-order system whose transfer function is in standard form as in Equation 10.7. Assume that the requirements for the system unit-step response are rise time \(t_{\mathrm{r}} \leq 0.1...
-
The transfer function of a dynamic system is given by \[G(s)=\frac{s+4}{s^{5}+2 s^{4}+3 s^{3}+8 s^{2}+4 s+5}\] Determine the stability of the system a. Using Routh's stability criterion without...
-
The unit-step response of a dynamic system is shown in Figure 10.12. Find the transfer function of the system if it can be approximated as \(a /\left(s^{2}+2 \zeta \omega_{n} s+\omega_{n}^{2}...

Study smarter with the SolutionInn App



