Explain the enthalpic and entropic contributions of the total contribution in Gibbs free energy, for the reaction
Fantastic news! We've Found the answer you've been seeking!
Question:
Explain the enthalpic and entropic contributions of the total contribution in Gibbs free energy, for the reaction process at B and C, shown in the figure below.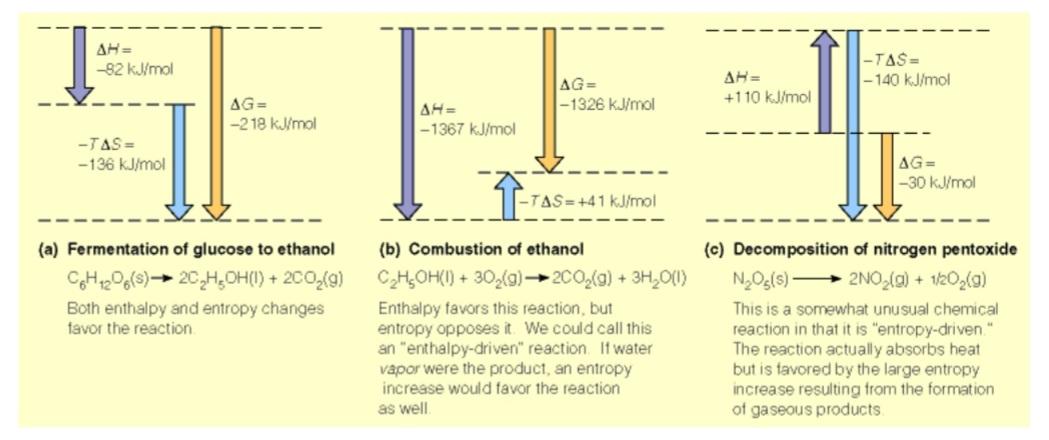
Transcribed Image Text:
AH= -TAS= -140 kJ/mol -82 k/mol AH= AG= -1326 kJ/mol +110 kJ/mol AG = -218 kJ/mol AH= -1367 kJ/mol -TAS = -136 KJ/mol AG= -30 kJ/mol -TAS=+41 kJ/mol (a) Fermentation of glucose to ethanol (b) Combustion of ethanol (c) Decomposition of nitrogen pentoxide CH206(s)→ 20,H,OH(1) + 200,(g) CH;OH(I) + 30,(g)200,(g) + 3H,O(0) NgOg(s) + 2NOz(g) + 1/20z(g) Enthalpy favors this reaction, but entropy opposes it We could call this an "enthalpy-driven" reaction If water vapor were the product, an entropy increase would favor the reaction Both enthalpy and entropy changes favor the reaction. This is a somewhat unusual chemical reaction in that it is "entropy-driven." The reaction actually absorbs heat but is tavored by the large entropy increase resulting from the formation of gaseous products as well. AH= -TAS= -140 kJ/mol -82 k/mol AH= AG= -1326 kJ/mol +110 kJ/mol AG = -218 kJ/mol AH= -1367 kJ/mol -TAS = -136 KJ/mol AG= -30 kJ/mol -TAS=+41 kJ/mol (a) Fermentation of glucose to ethanol (b) Combustion of ethanol (c) Decomposition of nitrogen pentoxide CH206(s)→ 20,H,OH(1) + 200,(g) CH;OH(I) + 30,(g)200,(g) + 3H,O(0) NgOg(s) + 2NOz(g) + 1/20z(g) Enthalpy favors this reaction, but entropy opposes it We could call this an "enthalpy-driven" reaction If water vapor were the product, an entropy increase would favor the reaction Both enthalpy and entropy changes favor the reaction. This is a somewhat unusual chemical reaction in that it is "entropy-driven." The reaction actually absorbs heat but is tavored by the large entropy increase resulting from the formation of gaseous products as well.
Expert Answer:
Answer rating: 100% (QA)
As we can see for all types of reactions G is negative but to make G negative the ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Explain how the free energy changes as a spontaneous reaction occurs. Show by means of a diagram how G changes with the extent of reaction.
-
Explain the relative distribution of products below using reaction energy diagrams for the hydrogen abstraction step that leads to each product. (The rate-determining step in radical halogenation is...
-
The standard free energy of activation of a reaction A is 90 kJ mol-1 (21.5 kcal mol-1) at 298 K. Reaction B is one million times faster than reaction A at the same temperature. The products of each...
-
Marsden Corp has developed a new strategic plan after rushing defective products to market hurt the company's sales and image. The strategic plan's initiatives are to focus on quality and develop...
-
What is the drawback of the default correlation measure and what alternative measure is used in measuring portfolio credit risk?
-
A waste treatment pond is 50m long and 15m wide, and has an average depth of 2m. The density of the waste is 85.3lb m /ft 3 . Calculate the weight of the pond contents in lbf, using a single...
-
On January 2, 2010, Shine Lights purchased showroom fixtures for $18,000 cash, expecting the fixtures to remain in service for five years. Shine Lights has depreci ated the fixtures on a...
-
The total factory overhead for Klein Calvin is budgeted for the year at $ 225,000, divided into four activities: cutting, $ 22,500; sewing, $ 45,000; setup, $ 100,000; and inspection, $ 57,500. Klein...
-
Q1) For all of this question: r = 5%, d = 1%, u = 1.07, d = 0.93 a) S = $56, K = $50, T = 3 months. Find the price of the call option with strike K expiring at time T by finding the replicating...
-
A welding torch is remotely controlled to achieve high accuracy while operating in changing and hazardous environments [21]. A model of the welding arm position control is shown in Figure DP7.4, with...
-
Labor Unions in the United States focus on objectives such as: A. Training their members for specific occupations B. Negotiations with corporate managers C. Organizing the local authorities D....
-
If the government grants exclusive rights for wheat production to two specific firms INDI and SRI instead of allowing widespread production across the country and competitive market. This...
-
Determine which details are facts and which are opinions. Write the letter of each detail in the matching column. Topic Sentence: The World's Fair was held in Chicago in 1893. a. The World's Fair was...
-
1. Graph y=(x + 1)
-
The purpose of this assignment is for students to explain the ethical obligations of attorneys to maintain client confidence. Course Competency: Apply ethical standards of conduct with clients,...
-
The discipline incident involves a 6th grade student named Travis and a fire in the boy's bathroom at school on December 12. Travis was suspected of starting the fire and on December 13, xxxx, the...
-
After repurchasing 5 % of its shares outstanding last year, a company has 1 1 4 million shares outstanding this year ( down from 1 2 0 million last year ) . Net income this year is $ 2 8 5 million...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Devise a method for the determination of the pH of an aqueous solution.
-
Use a phase diagram like that shown in Fig. 6.36 to indicate how zone leveling may be described.
-
What information does the term symbol F, provide about the angular momentum of an atom?
-
Design a voltage reference source as shown in Figure 9.42 to have an output voltage of \(12.0 \mathrm{~V}\). A Zener diode with a breakdown voltage of \(5.6 \mathrm{~V}\) is available. Assume the...
-
Design an op-amp summer to produce an output voltage of \(v_{O}=3 v_{I 1}+\) \(1.5 v_{I 2}+2 v_{I 3}-4 v_{I 4}-6 v_{I 5}\). The largest resistor value is to be \(250 \mathrm{k} \Omega\).
-
Consider the bridge circuit in Figure P9.87. The resistor \(R_{T}\) is a thermistor with values of \(20 \mathrm{k} \Omega\) at \(T=300 \mathrm{~K}\) and \(21 \mathrm{k} \Omega\) at \(T=350...

Study smarter with the SolutionInn App


