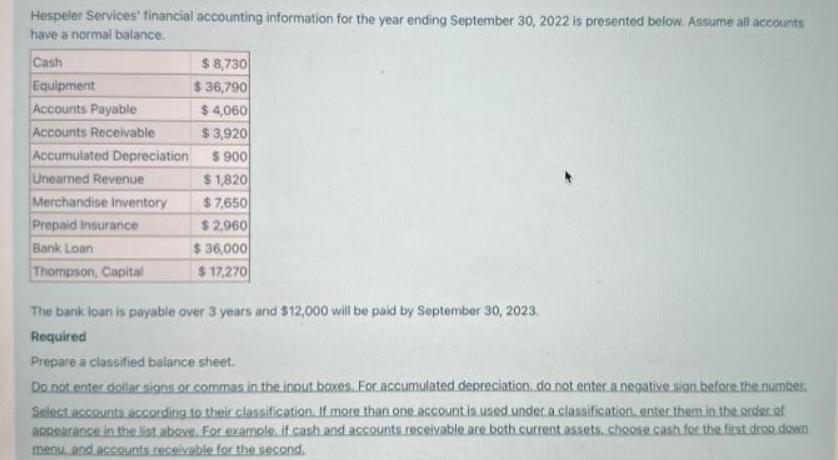
Hespeler Services financial accounting information for the year ending September 30, 2022 is presented below. Assume...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Hespeler Services financial accounting information for the year ending September 30, 2022 is presented below. Assume all accounts have a normal balance. Cash Equipment Accounts Payable Accounts Receivable Accumulated Depreciation Unearned Revenue Merchandise Inventory Prepaid Insurance Bank Loan Thompson, Capital $8,730 $36,790 $4,060 $3,920 $ 900 $1,820 $7,650 $ 2,960 $36,000 $17,270 The bank loan is payable over 3 years and $12,000 will be paid by September 30, 2023. Required Prepare a classified balance sheet. Do not enter dollar signs or commas in the input boxes. For accumulated depreciation, do not enter a negative sign before the number. Select accounts according to their classification. If more than one account is used under a classification, enter them in the order of appearance in the list above. For example, if cash and accounts receivable are both current assets. choose cash for the first drop down menu, and accounts receivable for the second. Hespeler Services financial accounting information for the year ending September 30, 2022 is presented below. Assume all accounts have a normal balance. Cash Equipment Accounts Payable Accounts Receivable Accumulated Depreciation Unearned Revenue Merchandise Inventory Prepaid Insurance Bank Loan Thompson, Capital $8,730 $36,790 $4,060 $3,920 $ 900 $1,820 $7,650 $ 2,960 $36,000 $17,270 The bank loan is payable over 3 years and $12,000 will be paid by September 30, 2023. Required Prepare a classified balance sheet. Do not enter dollar signs or commas in the input boxes. For accumulated depreciation, do not enter a negative sign before the number. Select accounts according to their classification. If more than one account is used under a classification, enter them in the order of appearance in the list above. For example, if cash and accounts receivable are both current assets. choose cash for the first drop down menu, and accounts receivable for the second.
Expert Answer:
Answer rating: 100% (QA)
Balance sheet as at March 20 Pl Cussent Assets Cash Accounts Receivable In... View the full answer

Related Book For 

Accounting Information Systems Controls and Processes
ISBN: 978-1119329565
3rd edition
Authors: Leslie Turner, Andrea Weickgenannt, Mary Kay Copeland
Posted Date:
Students also viewed these accounting questions
-
Calculate monthly returns, expected (average) returns and standard deviations of returns for each of the six stocks and the market index S&P/ASX300. Calculate correlation coefficients and covariance...
-
The FDA-approved RFID (Radio Frequency Identification) chips that can be implanted on clothes or even under the skin in humans. Some hospitals in Europe have begun implanting patients with RFID tags...
-
Coopers Cues Company is a small manufacturing operation that makes and sells pool cues for sporting goods stores and billiard halls in Baltimore, Maryland, and the surrounding local area. Rob Cooper...
-
Drag the labels onto the diagram to identify the cell types and matrix components of areolar connective tissue, a model connective tissue. Neutrophil Collagen fiber Lymphocyte Ground substance...
-
With respect to Test Yourself Question 1, suppose that wages rise to $20 per hour and glue prices rise to $6 per gallon. How are CCC's optimal input proportions likely to change?
-
How would you minimize the chances that stereotyping could affect the evaluation process in your company?
-
Taj Mahabub was the founder and CEO of GenAudio, a Colorado-based audio technology company. GenAudio had struggled financially practically since its formation, and Mahabub wished to secure a...
-
The following selected transactions were completed by Green Lawn Supplies Co., which sells irrigation supplies primarily to wholesalers and occasionally to retail customers: July 1. Sold merchandise...
-
When two electric charges are held a distance r apart, the electrostatic force between them is FE. The distance between the charges is then changed to 1r. (Enter numerical value only) The new...
-
The diagram shows the curve C with parametric equations The curve crosses the y-axis and the x-axis at points A and B respectively. The line l intersects the curve at points A and B. Find the...
-
Aluminum (E = 70 Gpa) workpiece CE is clamped in place as shown here. This piece is 2-cm-tall and has a 0.5 cm x 1 cm rectangular cross-section. A 4-cm-long- steel (E = 200 Gpa), 1-cm-diameter bolt...
-
What income tax effect does a partnership's receipt of tax-exempt interest and the proceeds of key-person life insurance payable on account of the death of the insured have upon the partnership...
-
Can a student attending a class in night school qualify as a full-time student?
-
How does a joint and survivor annuity differ from a single person annuity? How is the computation of the exclusion different for the joint annuity?
-
How do exchanging shareholders and security holders determine their basis for the stock and securities received in a corporate reorganization?
-
May a corporation receive an extension of the 2 1/2-month period for a retroactive S corporation election?
-
BMW's strategic analysis of the transition to electric vehicles. What alternatives are there for consumers and for the BWM to be successful?
-
Find i 0 (t) for t > 0 in the circuit in Fig. 16.72 . 2 + Vo 1 7.5e-2t u(t) V ( +) 4.5[1 u(t)]V 0.5v. 1H
-
Name the COSO reports five internal control activities.
-
Sensible CDs is a regional retail chain that sells used CDs. The company has eight stores throughout the Philadelphia region. At each store, customers can bring in used CDs to sell to Sensible or to...
-
Middleton Corporation just became a public corporation when shares of its stock were sold to the public three months ago. A new board of directors has been appointed to govern the corporation. Assume...
-
An ideal, isothermal, reversible fuel cell with reactants of oxygen and hydrogen, and a product of water operates at a temperature of \(400 \mathrm{~K}\) and a pressure of 1 bar. If the operating...
-
An electric cell has the following chemical reaction \[\mathrm{Zn}(\mathrm{s})+2 \mathrm{AgCl}(\mathrm{s})=\mathrm{ZnCl}_{2}+2 \mathrm{Ag}(\mathrm{s})\] and produces an emf of \(1.005 \mathrm{~V}\)...
-
A hydrogen-oxygen fuel cell is required to produce a constant voltage and operate over a pressure range of \(0.125-10\) bars. The datum voltage is \(1.16 \mathrm{~V}\) at a temperature of \(350...

Study smarter with the SolutionInn App


