f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid...
Fantastic news! We've Found the answer you've been seeking!
Question:
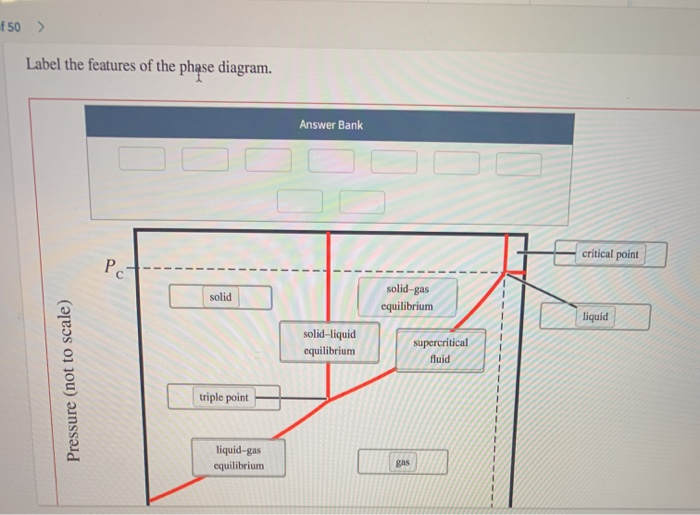
Transcribed Image Text:
f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid triple point liquid-gas equilibrium Answer Bank solid-liquid equilibrium solid-gas equilibrium gas supercritical fluid critical point liquid f 50 > Label the features of the phase diagram. Pressure (not to scale) Pc- solid triple point liquid-gas equilibrium Answer Bank solid-liquid equilibrium solid-gas equilibrium gas supercritical fluid critical point liquid
Expert Answer:
Answer rating: 100% (QA)
The lebels are correctly placed in the phase diagram of CO as follows Pressure not to scal... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The pressure of a gas at the triple point of water is 1.35 atm. If its volume remains unchanged, what will its pressure be at the temperature at which CO2 solidifies?
-
Label the regions of the phase diagram in Fig. 6.3 7. State what substances (if compounds give their formulas) exist in each region. Label each substance in each region as solid, liquid, or gas.
-
The equilibrium pressure of H, over solid uranium and uranium hydride, UH3' at 500 K is 139 Pa. Calculate the standard Gibbs energy of formation ofUH3 (s) at 500 K.
-
Maicom Construction Materials Inc. , hereinafter referred to as "MCM", is a construction materials company established in Moncton, New Brunswick. Its facilities (warehouse, store and offices) are...
-
Financial information for Buckle is presented in Appendix B at the end of the book. Required: 1. Determine whether the trend in net sales has been increasing or decreasing for the past three years....
-
Daniel Glaser, chairman of the College of San antonios business department, needs to assign professors to courses next semester. As a criterion for judging who should teach each course, Professor...
-
Im gathering some information about the sales/collection process and how it is supposed to work. Okay?
-
Compensated Absences Mathewson Company began operations on January 2, 2010. It employs 9 individuals who work 8-hour days and are paid hourly. Each employee earns 10 paid vacation days and 6 paid...
-
Given that R is the relation defined on TL as follows: "For every m, n = Z, m R n 3|(m-n). (a) Prove that the relation is an equivalence relation. (b) Identify the distinct equivalence classes of...
-
Suppose that it is one year after EBVs investment in Newco, and Tall-tree makes a Series B investment for 6M shares of Newco at $0.2 per share. Following the Series B investment, what percentage of...
-
A toy manufacturer produces two types of toy cars: sports cars and trucks. The sports cars require 3 units of plastic and 1 unit of metal to produce, while the trucks require 2 units of plastic and 2...
-
1 Identify the data collection methods used in each article. 1.1 Using examples from each article identify the data collection methods used in each study. Motivate each answer with examples from...
-
The range of the graph y=tan(0) is Write your answer using interval notation
-
3. Find the volume of the solid bounded above from above by the cone 2 = 4x+4y, bounded from below by the xy- plane and bounded laterally by the cylinder where a > 0. x+ y = ax.
-
Watch the TED Talk . Using your readings this week as a guide, apply what you've learned in the text by answering the following discussion questions (include references to textbook whenever...
-
We spent some time to understand what worldview is and looked at some historical and societal events that may have influenced our worldview. What is your worldview? Briefly share yours with the...
-
4 1 $97.25 2 $94.53 5 $87.53 $89.23 The above table shows the price per $100 face value of several risk-free, zero-coupon bonds. What is the yield to maturity of the three-year, zero-coupon,...
-
Players A, B, and C toss a fair coin in order. The first to throw a head wins. What are their respective chances of winning?
-
The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds: (a) N2O (b) NO (c) NO2 (d) N2O5 (e) N2O4.
-
Acetylene gas, C2H2 (g), can be prepared by the reaction of calcium carbide with water: CaC2 (s) + 2 H2O (l) Ca (OH)2 (s) + C2H2 (g) Calculate the volume of C2H2 that is collected over water at by...
-
A reaction that contributes to the depletion of ozone in the stratosphere is the direct reaction of oxygen atoms with ozone: At 298 K the rate constant for this reaction is 4.8 Ã 105 M-1 s-1....
-
Enter the following transactions of an antiques shop in the accounts and extract a trial balance as at 31 March 2017. 2017 March 1 Started in business with 8,000 in the bank. 2 Bought goods on time...
-
Record the following transactions for the month of January of a small finishing retailer, balance-off all the accounts, and then extract a trial balance as at 31 January 2016. 2016 Jan 1 Started in...
-
On 1 October 2016, the owner of the USS Enterprise, Mr Kirk, decides that he will boldly go and keep his records on a double entry system. His assets and liabilities at that date were: Kirk's...

Study smarter with the SolutionInn App


