Find the flame temperature of the products of combustion of hydrogen and oxygen whose composition and...
Fantastic news! We've Found the answer you've been seeking!
Question:
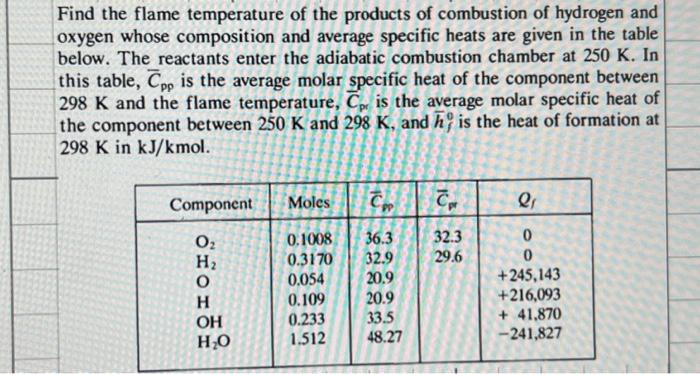
Transcribed Image Text:
Find the flame temperature of the products of combustion of hydrogen and oxygen whose composition and average specific heats are given in the table below. The reactants enter the adiabatic combustion chamber at 250 K. In this table, Cpp is the average molar specific heat of the component between 298 K and the flame temperature, Cp is the average molar specific heat of the component between 250 K and 298 K, and h, is the heat of formation at 298 K in kJ/kmol. Component 0₂ OH H₂O Moles C 0.1008 0.3170 0.054 0.109 0.233 1.512 36.3 32.9 20.9 20.9 33.5 48.27 Cpr 32.3 29.6 a Q₁ 0 0 +245,143 +216,093 +41,870 -241,827 Find the flame temperature of the products of combustion of hydrogen and oxygen whose composition and average specific heats are given in the table below. The reactants enter the adiabatic combustion chamber at 250 K. In this table, Cpp is the average molar specific heat of the component between 298 K and the flame temperature, Cp is the average molar specific heat of the component between 250 K and 298 K, and h, is the heat of formation at 298 K in kJ/kmol. Component 0₂ OH H₂O Moles C 0.1008 0.3170 0.054 0.109 0.233 1.512 36.3 32.9 20.9 20.9 33.5 48.27 Cpr 32.3 29.6 a Q₁ 0 0 +245,143 +216,093 +41,870 -241,827
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
Jim wants to allow a partner organization's Active Directory forest (B) to access his domain forest's (A)'s resources but doesn't want to allow users in his domain to access B's resources. He also...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Explain how to apply the rules of Section (61)a to determine taxable items.
-
Joel purchased 100 shares of stock for $20 per share. During the year, he received dividend cheques amounting to $150. After three years, Joel sold the stock for $32 per share. What was his holding...
-
Suppose that you are measuring the time to receive a TPDU. When an interrupt occurs, you read out the system clock in milliseconds. When the TPDU is fully processed, you read out the clock again. You...
-
During the past few years, Harry Davis Industries has been too constrained by the high cost of capital to make many capital investments. Recently, though, capital costs have been declining, and the...
-
Derive Equation 2.27. = S22 E S11S22 S2 1-V12V21 - S12 V12E2 SS22 S2 1-V12V21 S11 - S11S22-S2 1 = == S66 G12 = E2 1-V12V21 Q11 = Q12= Q22 Q66 = Q21 (2.27)
-
Refer to the information in Exercise 22 for Wyoming Wholesale. a. Using the least squares method, develop the equation for predicting weekly receiving report costs based on the number of shipments...
-
2 Pharoah Games Inc. adjusts its accounts annually. The following information is available for the year ended December 31, 2025. 1. Purchased a 1-year insurance policy on June 1 for $2,880 cash. 2....
-
It is now January 2018. The year 2017 finished well. The finalized year-end financial statements for 2017 are provided in the excel file associated with this assignment. Randolf and Tenisa at Comfy...
-
1. How might changes in the level of dividends cause conflicts of interest between holders of different kinds of the companys securities? 2. How does the discount on the price of shares sold via a...
-
A motorbike goes horizontally over a cliff. The cliff is 70m high. If the bikes velocity was 30km/h, how far from the base of the cliff does the car land?
-
1) Rim. Inc. , which had earnigns and profits of 100,000, distributed and to Alex Rowe, a stockholder Pym's adjusted basis for this land was $3,000. The land had a fair market value of 12,000 and was...
-
Ben Rishi is operations manager for a factory making saucepans. The weekly maximum capacity of the factory is 3000 units. The main limit on capacity is the old-fashioned machine for stamping out the...
-
31.Michael Company had 2,000 units in work in process at January 1 that were 80 percent complete. During January, 15,000 units were completed. At January 31, 4,000 units remained in work in process...
-
The Morgantown Power Plant burns oil, coal, and culm (waste from coal) to generate electricity. The plant burns 300 tons of coal, 398,750 tons of culm, and 248,470 gallons of oil. The plant generates...
-
Discuss methods of laying underground cables.
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Determine the change in the internal energy of helium, in kJ/kg, as it undergoes a change of state from 100 kPa and 20oC to 600 kPa and 300oC using the equation of state P(v - a) = RT where a = 0.01...
-
Repeat Prob. 15 -42 for liquid octane (C8H18). TABLE A-26 Enthalpy of formation, Gibbs function of formation, and absolute entropy at 25C, 1 atm Formula kJ/kmol J/k kJ/kmK Hydrogen 0 130.68 0 191.61...
-
The thrust developed by the engine of a Boeing 777 is about 380 kN. Assuming choked flow in the nozzles, determine the mass flow rate of air through the nozzle. Take the ambient conditions to be 220...
-
Find the probability that a randomly selected person has an IQ score higher than 125. Is this an unusual event? Explain. In a standardized IQ test, scores are normally distributed, with a mean score...
-
Find the probability that the number of U.S. adults who say they have had someone take over their email accounts without their permission is (a) at most 40, (b) less than 45, and (c) exactly 48....
-
A random sample of 60 people is selected from this population. What is the probability that the mean IQ score of the sample is greater than 105 ? Interpret the result. In a standardized IQ test,...

Study smarter with the SolutionInn App


