For a roadway project consisting of 40 kilometers, construct a line of balance diagram given that...
Fantastic news! We've Found the answer you've been seeking!
Question:
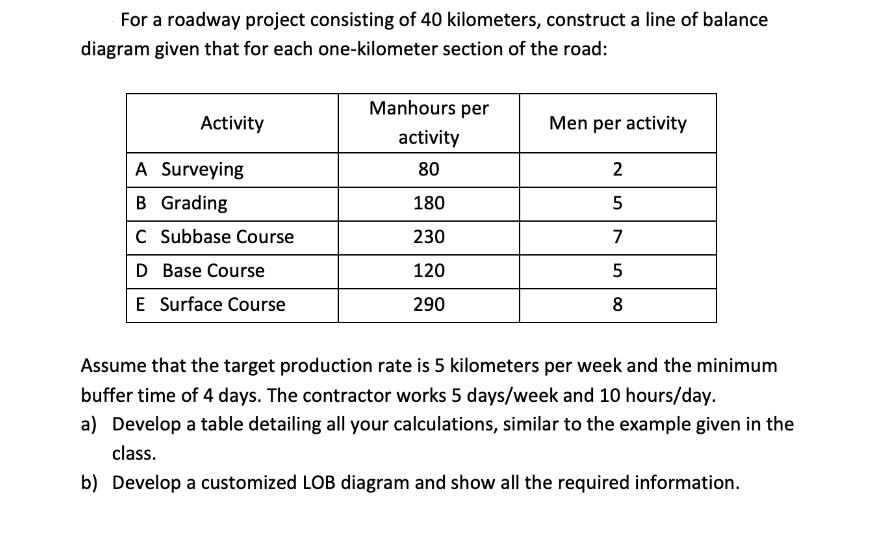
Transcribed Image Text:
For a roadway project consisting of 40 kilometers, construct a line of balance diagram given that for each one-kilometer section of the road: Activity A Surveying B Grading C Subbase Course D Base Course E Surface Course Manhours per activity 80 180 230 120 290 Men per activity 2 5 7 5 8 Assume that the target production rate is 5 kilometers per week and the minimum buffer time of 4 days. The contractor works 5 days/week and 10 hours/day. a) Develop a table detailing all your calculations, similar to the example given in the class. b) Develop a customized LOB diagram and show all the required information. For a roadway project consisting of 40 kilometers, construct a line of balance diagram given that for each one-kilometer section of the road: Activity A Surveying B Grading C Subbase Course D Base Course E Surface Course Manhours per activity 80 180 230 120 290 Men per activity 2 5 7 5 8 Assume that the target production rate is 5 kilometers per week and the minimum buffer time of 4 days. The contractor works 5 days/week and 10 hours/day. a) Develop a table detailing all your calculations, similar to the example given in the class. b) Develop a customized LOB diagram and show all the required information.
Expert Answer:
Answer rating: 100% (QA)
Answer a Table of Calculations Section Activity Manhours Men Required Days Required Start Date End D... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these general management questions
-
Smythe Co. makes furniture. The following data are taken from its production plans for the year. Direct labor costs $5,720,000 Hazardous waste disposal costs 620,000 Chairs Expected production...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The registrar at a prominent northeastern university recently scheduled an instructor to teach two different classes at the same exact time. Help the registrar prevent future mistakes by describing a...
-
Ven Company is a retailer. In 2016, its before-tax net income for financial reporting purposes was $600,000. This included a $150,000 gain from the sale of land held for several years as a possible...
-
The accounting records of Jim's Appliances included the following balances at the end of the period: In the past, Jim's warranty expense has been 9% of sales. During the current period, the business...
-
The June 30, 2010, adjusted trial balance of Energized Espresso, Inc., is shown next. Requirements 1. Prepare the June closing entries for Energized Espresso, Inc. 2. Calculate the ending balance in...
-
Variable costing versus absorption costing Lange Company incurred manufacturing overhead cost for the year as follows. The company produced 1,500 units and sold 1,000 of them at $180 per unit. Assume...
-
B ] The equity section of SKM Corporation's statement of financial position as of December 3 1 , 2 0 1 2 is as follows: Equity Share capital ordinary, $ 5 par value; authorized, 2 , 0 0 0 , 0 0 0...
-
Stainless Steel Specialties (SSS) is a manufacturer of hot water-based heating systems for homes and commercial businesses. The company has grown about 10% in each of the past five years. The company...
-
If you are planning for a negotiation presentation with an investor and you know that the individual is a Director (or Directive) personality-communication type, how would you plan your presentation?
-
You are trying to measure the width of a sidewalk, but you measure the distances from the street to each side of the sidewalk. If you measure the far side of the sidewalk to be 24.30.05 m from the...
-
In the program below, of the two lines of code with a comment, which one is not allowed (i.e., would result in an error)? int main() { int *ptr; int a[2], b[2]; ptr = a; // line 4 b = a; // line 5 }
-
Warnerwoods Company uses a perpetual inventory system. It entered into the following purchases and sales transactions for March. Date March 1 Activities Beginning inventory Units Acquired at Cost...
-
Using properties of proportion find xy, given: x + 2x 2x + 4 = y+ 3y 3y+9
-
In the manufacture of 9,200 units of a product, direct materials cost incurred was $173,900, direct labor cost incurred was $111,600, and applied factory overhead was $49,000. The total conversion...
-
Consider X-ray diffraction from a crystal with face centred cubic (fcc) lattice. The lattice plane(s) for which diffraction peaks can be observed is/are: (2, 1, 2) (1, 1, 1) (2, 0, 0) (3, 1, 1)
-
Write a paper about how diet relates to breast cancer in women study design to use: case control study purpose & rationale the purpose of this final project is to utilize the methods and...
-
Ulysses and Penelope are married and file separate returns for 2012. Penelope itemizes her deductions on her return. Ulysses' adjusted gross income was $17,400, his itemized deductions were $2,250,...
-
Sally and Charles Heck received the following dividends and interest during 2012: Assuming the Hecks file a joint tax return, complete Schedule B of Form 1040 (on page 2-33) for them for the 2012 tax...
-
In June of 2012, Maureen's house is vandalized during a long-term power failure after a hurricane hit the city. The president of the United States declares Maureen's city a disaster area as a result...
-
Show that for an ideal gas, \(\left(\frac{\partial E}{\partial V} ight)_{T}=0\), and for a van der Waals gas, \(\left(\frac{\partial E}{\partial V} ight)_{T}=\frac{a n^{2}}{V^{2}}\).
-
Explain some important methods for the estimation of the fugacity coefficient of a pure substance.
-
Estimate the residual entropy, enthalpy and internal energy at 298 K and 10 bar for nitrogen obeying the van der Waals equation of state, given that T c = 126.2 K and P c = 34.0 bar.

Study smarter with the SolutionInn App


