For each of the following rules for the function f: N N. make a table to...
Fantastic news! We've Found the answer you've been seeking!
Question:

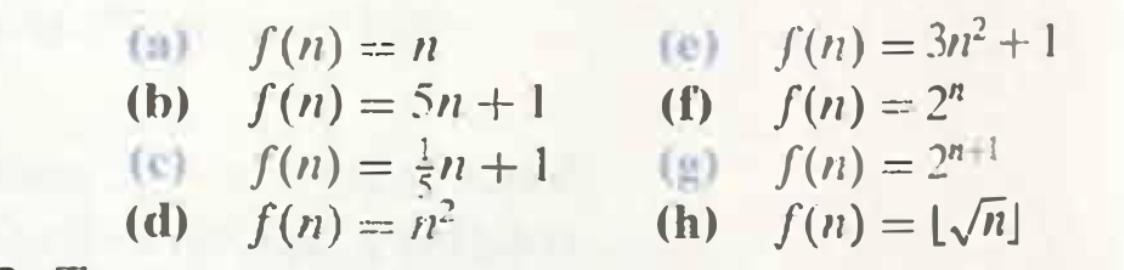

Transcribed Image Text:
For each of the following rules for the function f: N→ N. make a table to investigate the ratio S(2n)/ f(n) for increasing values of n. For each definition of S. write a sentence of the form "When the input value is doubled, the output is (a) S(n) = n (b) S(n) = 5n+1 (c) f(n) = n+ 1 (d) f(n) = n² (e) S(n) = 3² + 1 (f) f(n) = 2* (g) S(n) = 2"+1 (h) f(n) = [/n] From Section 4.8: • Exercise 2. See partial answers in book to see what kind of answer we're looking for. A reasonable format for the inspection table for each part would be something like this: f(n) f(2n) f(2n)/ f(n) 10 20 40 For each of the following rules for the function f: N→ N. make a table to investigate the ratio S(2n)/ f(n) for increasing values of n. For each definition of S. write a sentence of the form "When the input value is doubled, the output is (a) S(n) = n (b) S(n) = 5n+1 (c) f(n) = n+ 1 (d) f(n) = n² (e) S(n) = 3² + 1 (f) f(n) = 2* (g) S(n) = 2"+1 (h) f(n) = [/n] From Section 4.8: • Exercise 2. See partial answers in book to see what kind of answer we're looking for. A reasonable format for the inspection table for each part would be something like this: f(n) f(2n) f(2n)/ f(n) 10 20 40
Expert Answer:
Answer rating: 100% (QA)
1 solution Lets find first all fans for respective fin Ju... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Write a balanced equation for each of the following reactions: (a) Sulfur dioxide reacts with water. (b) Solid zinc sulfide reacts with hydrochloric acid. (c) Elemental sulfur reacts with sulfite ion...
-
Write a structural formula for each of the following compounds: (a) 6-Isopropyl-2, 3-dimethylnonane (e) Cyclobutylcyclopentane (b) 4-tert-Butyl-3-methylheptane (f) (2, 2-Dimethylpropyl) cyclohexane...
-
Write structural formulas for each of the following bicyclic hydrocarbons: (a) Bicyclo [2.2.1] heptane (c) Bicyclo [3.1.1] heptane (b) Bicyclo [5.2.0] nonane (d) Bicyclo [3.3.0] octane
-
Given the following data: Calculate ÎH for the reaction On the basis of enthalpy change, is this a useful reaction for the synthesis of ammonia? AH - 92 kJ () + AH = -484 k (g) ON OH 88
-
A firm has a monthly production function where L is hours of labor per month and K is square feet of manufacturing space. The marginal product of labor is The marginal product of capital is a. If the...
-
Which of the following accounts normally has a debit balance? a. Unearned Revenue b. Rent Expense c. Retained Earnings d. Sales Revenue
-
Very high vibration levels were experienced on a drag line when the bucket, full of wet sand, was shifted into high speed for the ascent from the lake. The frequency of this peak is much lower than...
-
Several specific audit procedures are listed below. For each item, identify the type of procedure listed and which of the assertions is being addressed by the procedure. (a) Examine a list of...
-
A company is considering two mutually exclusive projects. The projected cash flows are as follows: Cash Flows:A Cash Flows:B Year 0 -$245,000 -$250,000 1 $70,500 $60,000 2 $85,000 $60,000 3 $90,000...
-
Michael and Jeanette Boyds Tax Return Michael D. and Jeanette S. Boyd live with their family at the Rock Glen House Bed & Breakfast, which Michael operates. The Bed & Breakfast (B&B) is...
-
What social media practices in the United States are fueling a pre-totalitarian culture? Explain
-
Briefly compare the coupon rate and the interest rate regarding bonds. What is a par value? Describe the impact of a tax shield on fixed income yields.
-
Take the cultural intelligence assessment. What does that tell you about yourself? What does that tell you about the differences between your culture and others?
-
Consider the simple regression model: Y = Bo+BX; +u; (a) Under the Classical Linear Regression Model assumptions, the ordinary least squares (OLS) estimators, Bo and B, are "unbiased." Explain. (b)...
-
Industry characteristics, firm characteristics, sales growth, profit margin, dividend policy, asset requirement, and leverage. How do these key factors affect forecasting of financial statements?
-
Primax Berhad had just paid divided of RM 0 . 4 4 . Through a new technique of producing their products, Primax expects to obtain high growth achievement in the short term that is at 2 5 percent per...
-
What cultural significance do indigenous folk art traditions hold within their respective communities, and how have they evolved over time amidst modernization and globalization?
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
The solubility of nitrogen in water is 8.21 10-4 mol/ L at 0oC when the N2 pressure above water is 0.790 atm. Calculate the Henrys law constant for N2 in units of L atm/mol for Henrys law in the...
-
Write balanced equations describing the reaction of Sr with each of the following: O2, S8, Cl2, P4, H2, H2O, and HCl.
-
When M 2 S 3 (s) is heated in air, it is converted to MO 2 (s). A 4.000- g sample of M 2 S 3 (s) shows a decrease in mass of 0.277 g when it is heated in air. What is the average atomic mass of M?
-
Cooling pipes at three nuclear power plants are investigated for deposits that would inhibit the flow of water. From 30 randomly selected spots at each plant, 13 from the first plant, 8 from the...
-
With reference to Exercise 10.57, find a large sample 95% confidence interval for the true difference of the probabilities of failure. Data From Exercise 10.57 10.57 Two bonding agents, A and B, are...
-
Two hundred tires of each of four brands are individually placed in a testing apparatus and run until failure. The results are obtained the results shown in the following table: (a) Use the 0.01...

Study smarter with the SolutionInn App


