For jet aircraft flying out of Houston on a summer afternoon (T = 95 F, 70%...
Fantastic news! We've Found the answer you've been seeking!
Question:

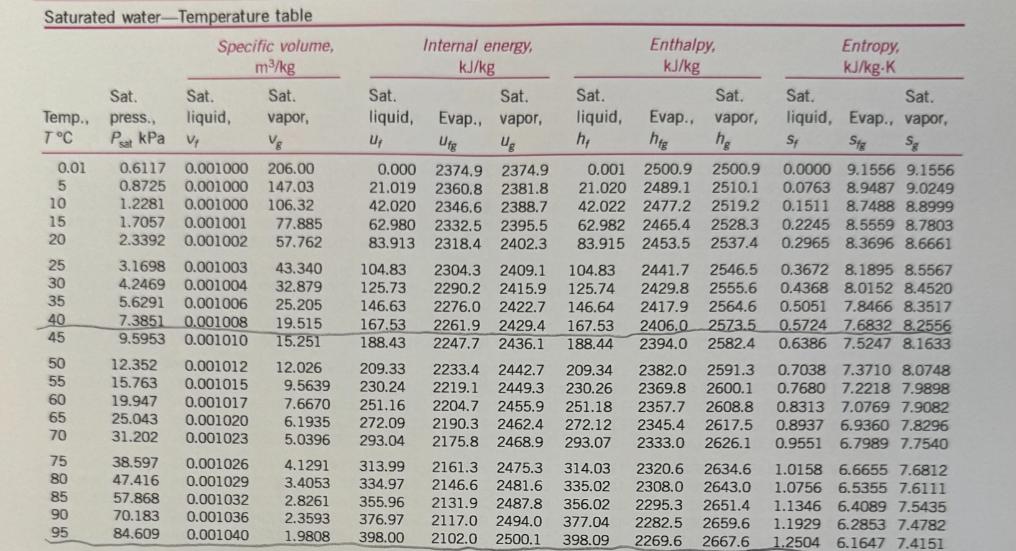
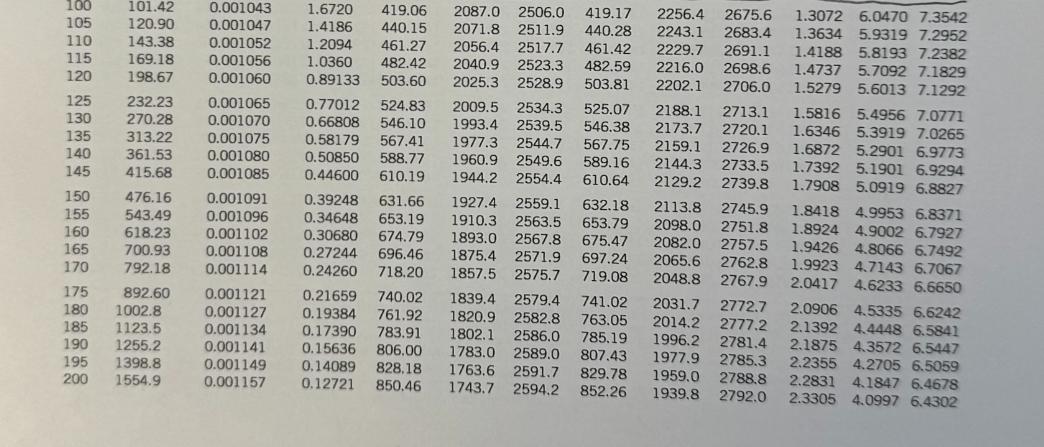
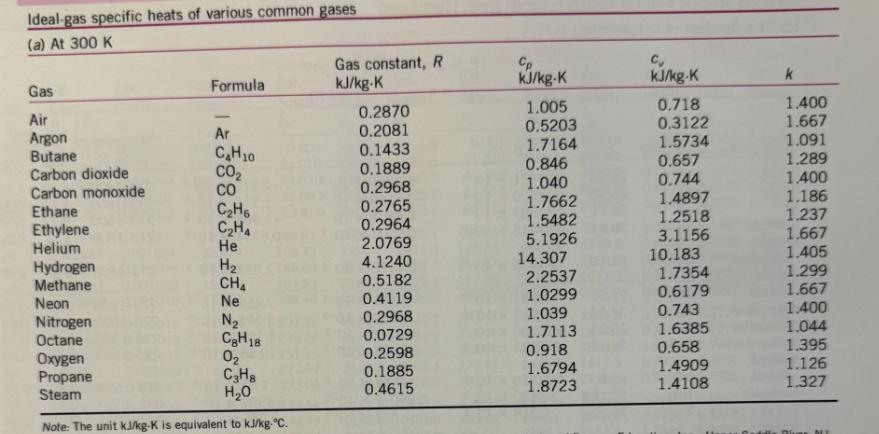
Transcribed Image Text:
For jet aircraft flying out of Houston on a summer afternoon (T = 95 °F, 70% relative humidity) and Palm Springs on a winter morning (T = 40 °F, 55% relative humidity), determine for both locations: a) the mass of water vapor in 1 m³ of air b) the thermal efficiency of an ideal Brayton cycle with a pressure ratio of 9. Assume that both cities are at sea level and both air and H₂O can be treated as ideal gases with constant specific heats evaluated at 300 K. Saturated water-Temperature table Specific volume, m³/kg Sat. Sat. Sat. Temp., press., liquid, vapor, T°C℃ Psat kPa V₂ Vg 0.01 BARR BARN4 888 5 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 0.6117 0.001000 206.00 0.8725 0.001000 147.03 1.2281 0.001000 106.32 1.7057 0.001001 77.885 2.3392 0.001002 57.762 3.1698 0.001003 43.340 4.2469 0.001004 32.879 5.6291 0.001006 25.205 7.3851 0.001008 19.515 9.5953 0.001010 15.251 12.352 15.763 19.947 25.043 31.202 0.001012 12.026 0.001015 0.001017 0.001020 0.001023 38.597 0.001026 47.416 0.001029 57.868 0.001032 70.183 0.001036 84.609 0.001040 Internal energy, kJ/kg Sat. liquid, Evap., Ufg U₁ 209.33 9.5639 230.24 7.6670 251.16 272.09 293.04 6.1935 5.0396 4.1291 313.99 3.4053 334.97 2.8261 355.96 2.3593 376.97 1.9808 398.00 Sat. vapor, Ug Sat. liquid, h₂ Enthalpy, kJ/kg Sat. Evap., vapor, hfg hg Entropy, kJ/kg-K 0.001 2500.9 2500.9 21.020 2489.1 2510.1 42.022 2477.2 2519.2 62.982 2465.4 2528.3 83.915 2453.5 2537.4 0.000 2374.9 2374.9 21.019 2360.8 2381.8 42.020 2346.6 2388.7 62.980 2332.5 2395.5 83.913 2318.4 2402.3 104.83 2304.3 2409.1 104.83 2441.7 2546.5 125.73 2290.2 2415.9 125.74 2429.8 2555.6 146.63 2276.0 2422.7 146.64 2417.9 2564.6 167.53 2261.9 2429.4 167.53 2406.0 2573.5 188.43 2247.7 2436.1 188.44 2394.0 2582.4 2233.4 2442.7 209.34 2382.0 2591.3 0.7038 7.3710 8.0748 2219.1 2449.3 230.26 2369.8 2600.1 0.7680 7.2218 7.9898 2204.7 2455.9 251.18 2357.7 2608.8 0.8313 7.0769 7.9082 2190.3 2462.4 272.12 2345.4 2617.5 0.8937 6.9360 7.8296 2175.8 2468.9 293.07 2333.0 2626.1 0.9551 6.7989 7.7540 2161.3 2475.3 314.03 2320.6 2634.6 1.0158 6.6655 7.6812 2146.6 2481.6 335.02 2308.0 2643.0 1.0756 6.5355 7.6111 2131.9 2487.8 356.02 2295.3 2651.4 2117.0 2494.0 377.04 2282.5 2659.6 2102.0 2500.1 398.09 2269.6 2667.6 1.1346 6.4089 7.5435 1.1929 6.2853 7.4782 1.2504 6.1647 7.4151 Sat. Sat. liquid, Evap., vapor, St Sig Sg 0.0000 9.1556 9.1556 0.0763 8.9487 9.0249 0.1511 8.7488 8.8999 0.2245 8.5559 8.7803 0.2965 8.3696 8.6661 0.3672 8.1895 8.5567 0.4368 8.0152 8.4520 0.5051 7.8466 8.3517 0.5724 7.6832 8.2556 0.6386 7.5247 8.1633 100 105 110 115 120 125 130 135 140 145 101.42 120.90 0.001043 0.001047 0.001052 169.18 0.001056 143.38 198.67 0.001060 232.23 270.28 0.001065 0.001070 0.001075 0.001080 415.68 0.001085 313.22 361.53 160 150 476.16 0.001091 155 543.49 0.001096 618.23 0.001102 165 700.93 0.001108 170 792.18 0.001114 175 892.60 0.001121 180 1002.8 0.001127 185 1123.5 190 1255.2 0.001134 0.001141 0.001149 0.001157 195 1398.8 200 1554.9 1.6720 419.06 2087.0 2506.0 419.17 2256.4 2675.6 1.4186 440.15 2071.8 2511.9 440.28 2243.1 2683.4 1.2094 461.27 2056.4 2517.7 461.42 2229.7 2691.1 1.0360 482.42 2040.9 2523.3 482.59 2216.0 2698.6 0.89133 503.60 2025.3 2528.9 503.81 2202.1 2706.0 2009.5 2534.3 525.07 2188.1 2713.1 1993.4 2539.5 546.38 2173.7 2720.1 1977.3 2544.7 567.75 2159.1 2726.9 1960.9 2549.6 589.16 2144.3 2733.5 1944.2 2554.4 610.64 2129.2 2739.8 1927.4 2559.1 632.18 2113.8 2745.9 1910.3 2563.5 653.79 2098.0 2751.8 1893.0 2567.8 675.47 2082.0 2757.5 1875.4 2571.9 697.24 2065.6 2762.8 1857.5 2575.7 719.08 2048.8 2767.9 1839.4 2579.4 741.02 2031.7 2772.7 1820.9 2582.8 763.05 2014.2 2777.2 1802.1 2586.0 785.19 1996.2 2781.4 1783.0 2589.0 807.43 1977.9 2785.3 1763.6 2591.7 829.78 1959.0 2788.8 1743.7 2594.2 852.26 1939.8 2792.0 0.77012 524.83 0.66808 546.10 0.58179 567.41 0.50850 588.77 0.44600 610.19 0.39248 631.66 0.34648 653.19 0.30680 674.79 0.27244 696.46 0.24260 718.20 0.21659 740.02 0.19384 761.92 0.17390 783.91 0.15636 806.00 0.14089 828.18 0.12721 850.46 1.3072 6.0470 7.3542 1.3634 5.9319 7.2952 1.4188 5.8193 7.2382 1.4737 5.7092 7.1829 1.5279 5.6013 7.1292 1.5816 5.4956 7.0771 1.6346 5.3919 7.0265 1.6872 5.2901 6.9773 1.7392 5.1901 6.9294 1.7908 5.0919 6.8827 1.8418 4.9953 6.8371 1.8924 4.9002 6.7927 1.9426 4.8066 6.7492 1.9923 4.7143 6.7067 2.0417 4.6233 6.6650 2.0906 4.5335 6.6242 2.1392 4.4448 6.5841 2.1875 4.3572 6.5447 2.2355 4.2705 6.5059 2.2831 4.1847 6.4678 2.3305 4.0997 6.4302 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Formula Oxygen Propane Steam Ar C₂H10 CO₂ CO C₂H6 C₂H4 He H₂ CH₂ Ne N₂ CH18 0₂ C3H8 H₂O Note: The unit kJ/kg-K is equivalent to kj/kg-°C. Gas constant, R kJ/kg-K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg-K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 Cu kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 NE For jet aircraft flying out of Houston on a summer afternoon (T = 95 °F, 70% relative humidity) and Palm Springs on a winter morning (T = 40 °F, 55% relative humidity), determine for both locations: a) the mass of water vapor in 1 m³ of air b) the thermal efficiency of an ideal Brayton cycle with a pressure ratio of 9. Assume that both cities are at sea level and both air and H₂O can be treated as ideal gases with constant specific heats evaluated at 300 K. Saturated water-Temperature table Specific volume, m³/kg Sat. Sat. Sat. Temp., press., liquid, vapor, T°C℃ Psat kPa V₂ Vg 0.01 BARR BARN4 888 5 10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 0.6117 0.001000 206.00 0.8725 0.001000 147.03 1.2281 0.001000 106.32 1.7057 0.001001 77.885 2.3392 0.001002 57.762 3.1698 0.001003 43.340 4.2469 0.001004 32.879 5.6291 0.001006 25.205 7.3851 0.001008 19.515 9.5953 0.001010 15.251 12.352 15.763 19.947 25.043 31.202 0.001012 12.026 0.001015 0.001017 0.001020 0.001023 38.597 0.001026 47.416 0.001029 57.868 0.001032 70.183 0.001036 84.609 0.001040 Internal energy, kJ/kg Sat. liquid, Evap., Ufg U₁ 209.33 9.5639 230.24 7.6670 251.16 272.09 293.04 6.1935 5.0396 4.1291 313.99 3.4053 334.97 2.8261 355.96 2.3593 376.97 1.9808 398.00 Sat. vapor, Ug Sat. liquid, h₂ Enthalpy, kJ/kg Sat. Evap., vapor, hfg hg Entropy, kJ/kg-K 0.001 2500.9 2500.9 21.020 2489.1 2510.1 42.022 2477.2 2519.2 62.982 2465.4 2528.3 83.915 2453.5 2537.4 0.000 2374.9 2374.9 21.019 2360.8 2381.8 42.020 2346.6 2388.7 62.980 2332.5 2395.5 83.913 2318.4 2402.3 104.83 2304.3 2409.1 104.83 2441.7 2546.5 125.73 2290.2 2415.9 125.74 2429.8 2555.6 146.63 2276.0 2422.7 146.64 2417.9 2564.6 167.53 2261.9 2429.4 167.53 2406.0 2573.5 188.43 2247.7 2436.1 188.44 2394.0 2582.4 2233.4 2442.7 209.34 2382.0 2591.3 0.7038 7.3710 8.0748 2219.1 2449.3 230.26 2369.8 2600.1 0.7680 7.2218 7.9898 2204.7 2455.9 251.18 2357.7 2608.8 0.8313 7.0769 7.9082 2190.3 2462.4 272.12 2345.4 2617.5 0.8937 6.9360 7.8296 2175.8 2468.9 293.07 2333.0 2626.1 0.9551 6.7989 7.7540 2161.3 2475.3 314.03 2320.6 2634.6 1.0158 6.6655 7.6812 2146.6 2481.6 335.02 2308.0 2643.0 1.0756 6.5355 7.6111 2131.9 2487.8 356.02 2295.3 2651.4 2117.0 2494.0 377.04 2282.5 2659.6 2102.0 2500.1 398.09 2269.6 2667.6 1.1346 6.4089 7.5435 1.1929 6.2853 7.4782 1.2504 6.1647 7.4151 Sat. Sat. liquid, Evap., vapor, St Sig Sg 0.0000 9.1556 9.1556 0.0763 8.9487 9.0249 0.1511 8.7488 8.8999 0.2245 8.5559 8.7803 0.2965 8.3696 8.6661 0.3672 8.1895 8.5567 0.4368 8.0152 8.4520 0.5051 7.8466 8.3517 0.5724 7.6832 8.2556 0.6386 7.5247 8.1633 100 105 110 115 120 125 130 135 140 145 101.42 120.90 0.001043 0.001047 0.001052 169.18 0.001056 143.38 198.67 0.001060 232.23 270.28 0.001065 0.001070 0.001075 0.001080 415.68 0.001085 313.22 361.53 160 150 476.16 0.001091 155 543.49 0.001096 618.23 0.001102 165 700.93 0.001108 170 792.18 0.001114 175 892.60 0.001121 180 1002.8 0.001127 185 1123.5 190 1255.2 0.001134 0.001141 0.001149 0.001157 195 1398.8 200 1554.9 1.6720 419.06 2087.0 2506.0 419.17 2256.4 2675.6 1.4186 440.15 2071.8 2511.9 440.28 2243.1 2683.4 1.2094 461.27 2056.4 2517.7 461.42 2229.7 2691.1 1.0360 482.42 2040.9 2523.3 482.59 2216.0 2698.6 0.89133 503.60 2025.3 2528.9 503.81 2202.1 2706.0 2009.5 2534.3 525.07 2188.1 2713.1 1993.4 2539.5 546.38 2173.7 2720.1 1977.3 2544.7 567.75 2159.1 2726.9 1960.9 2549.6 589.16 2144.3 2733.5 1944.2 2554.4 610.64 2129.2 2739.8 1927.4 2559.1 632.18 2113.8 2745.9 1910.3 2563.5 653.79 2098.0 2751.8 1893.0 2567.8 675.47 2082.0 2757.5 1875.4 2571.9 697.24 2065.6 2762.8 1857.5 2575.7 719.08 2048.8 2767.9 1839.4 2579.4 741.02 2031.7 2772.7 1820.9 2582.8 763.05 2014.2 2777.2 1802.1 2586.0 785.19 1996.2 2781.4 1783.0 2589.0 807.43 1977.9 2785.3 1763.6 2591.7 829.78 1959.0 2788.8 1743.7 2594.2 852.26 1939.8 2792.0 0.77012 524.83 0.66808 546.10 0.58179 567.41 0.50850 588.77 0.44600 610.19 0.39248 631.66 0.34648 653.19 0.30680 674.79 0.27244 696.46 0.24260 718.20 0.21659 740.02 0.19384 761.92 0.17390 783.91 0.15636 806.00 0.14089 828.18 0.12721 850.46 1.3072 6.0470 7.3542 1.3634 5.9319 7.2952 1.4188 5.8193 7.2382 1.4737 5.7092 7.1829 1.5279 5.6013 7.1292 1.5816 5.4956 7.0771 1.6346 5.3919 7.0265 1.6872 5.2901 6.9773 1.7392 5.1901 6.9294 1.7908 5.0919 6.8827 1.8418 4.9953 6.8371 1.8924 4.9002 6.7927 1.9426 4.8066 6.7492 1.9923 4.7143 6.7067 2.0417 4.6233 6.6650 2.0906 4.5335 6.6242 2.1392 4.4448 6.5841 2.1875 4.3572 6.5447 2.2355 4.2705 6.5059 2.2831 4.1847 6.4678 2.3305 4.0997 6.4302 Ideal-gas specific heats of various common gases (a) At 300 K Gas Air Argon Butane Carbon dioxide Carbon monoxide Ethane Ethylene Helium Hydrogen Methane Neon Nitrogen Octane Formula Oxygen Propane Steam Ar C₂H10 CO₂ CO C₂H6 C₂H4 He H₂ CH₂ Ne N₂ CH18 0₂ C3H8 H₂O Note: The unit kJ/kg-K is equivalent to kj/kg-°C. Gas constant, R kJ/kg-K 0.2870 0.2081 0.1433 0.1889 0.2968 0.2765 0.2964 2.0769 4.1240 0.5182 0.4119 0.2968 0.0729 0.2598 0.1885 0.4615 Cp kJ/kg-K 1.005 0.5203 1.7164 0.846 1.040 1.7662 1.5482 5.1926 14.307 2.2537 1.0299 1.039 1.7113 0.918 1.6794 1.8723 Cu kJ/kg-K 0.718 0.3122 1.5734 0.657 0.744 1.4897 1.2518 3.1156 10.183 1.7354 0.6179 0.743 1.6385 0.658 1.4909 1.4108 k 1.400 1.667 1.091 1.289 1.400 1.186 1.237 1.667 1.405 1.299 1.667 1.400 1.044 1.395 1.126 1.327 NE
Expert Answer:
Answer rating: 100% (QA)
Part a Mass of Water Vapor in 1 m of Air Houston Temperature conversion T1 95F 35C 308 K Vapor press... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these mechanical engineering questions
-
Estimate the Slope (Slope = Risefsun) of the tangent line to the cure. What is your estimate of the slope? (Round to the nearest intreges) Slope
-
4. Read the article (posted) from September 20th 2023 titled "Fed Aims for Soft Landing With a Rate Pause and a Pledge to Move Carefully" (a) What does soft landing" mentioned in the article refer...
-
An ideal Brayton cycle with regeneration has a pressure ratio of 10. Air enters the compressor at 300 K and the turbine at 1200 K. If the effectiveness of the regenerator is 100 percent, determine...
-
When Ralph Lauren makes shirts to a customers exact preferences, what utility is provided?
-
The owner of a large fleet of taxis is trying to estimate his costs for next year's operations. One major cost is fuel purchases. To estimate fuel purchases, the owner needs to know the total...
-
Estimate the largest value of x that allows e x to be approximated to 1% accuracy by the following partial sum: e x 1 + x.
-
Why do you think that people, on average, choose to have fewer children than they did a generation ago? Can anything be done to reverse the trend? How far should governments see it as their role to...
-
Only one firm produces and sells soccer balls in the country of Wiknam, and as the story begins, international trade in soccer balls is prohibited. The following equations describe the monopolists...
-
Mr. and Mrs. Twig are both under 65 years of age and have no dependents. Their only income for the year was his salary of $45,500. During the year they made only a nominal amount of disbursements of...
-
Discuss the use of business analytics in sports, such as tennis, cricket, swimming, and football. Identify as many opportunities as you can for each.
-
An action plan for a personal leadership portfolio. Please don't forget support in-text as well as in references. Developing the degree of self-awareness and direction setting as this assignment...
-
Given below is some is a comparison of financial performance data of a project when flexibility is incorporated (I.e. flexible project) in comparison to when it is not. (i.e. inflexible project) The...
-
For Service Zone H, assuming your shipment chargeable weight is between 100 and 300 kg, at what weight does it become cheaper to declare the shipment weight to be 300 kg.? EG: What is the rate break...
-
Gold Dust Ltd has produced the following budgeted data for its current financial year:- Sales 2900000 Direct materials 400000 Direct labour 500000 Production overhead 1200000 Production cost 2100000...
-
Critical Review V Hide Assignment Information Instructions Williams, A. (2012). Worry, intolerance of uncertainty, and statistics anxiety. Click on the following link to retrieve the article....
-
(4.) Octopussy Company uses a predetermined overhead rate in applying overhead to production orders on a labor-cost basis for Dept. A and on a machine-hour basis for Dept. B. At the beginning of...
-
Assuming a discount rate of 6 percent, which of the following has the highest cash value (present value at the date of purchase)? Assume all purchase options below are as at the same date. There will...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
When you wind a spring up in a toy or stretch a rubber band what happens in terms of work, energy and heat transfer? Later when they are released, what happens then?
-
The reheat pressure effect the operating variables and thus turbine performance. Repeat Problem 11.37 twice, using 0.6 and 1.0 MPa for the reheat pressure.
-
Consider the steam turbine in Example 6.6. Is this a reversible process?
-
How would the value chain approach to internal analysis be used? (R-56)
-
Does this seem appropriate for the situation the organization is in now?
-
Does the culture appear to be strong or weak?

Study smarter with the SolutionInn App


