distillation, a number of methods were proposed for solving the equations graphically or algebraically during an...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
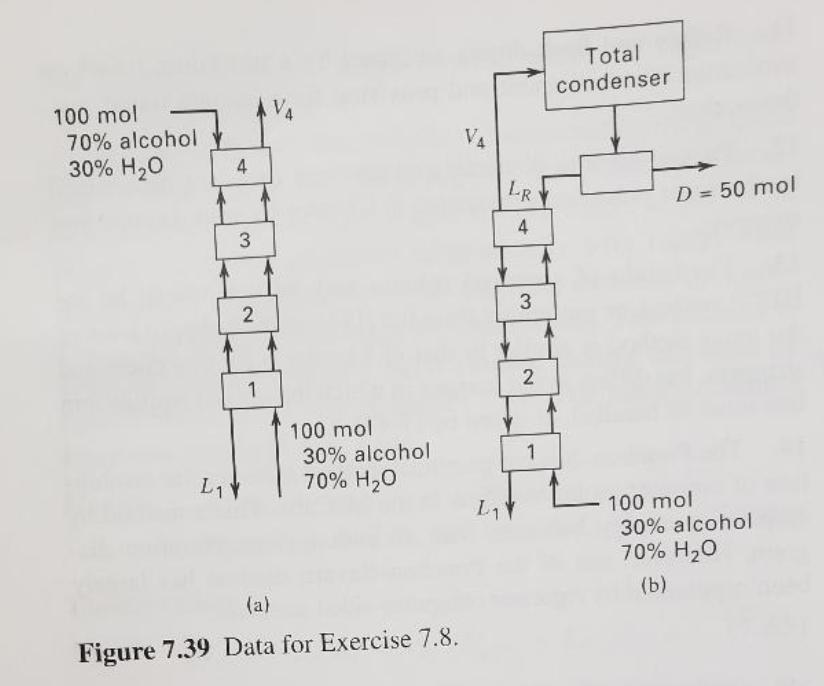
distillation, a number of methods were proposed for solving the equations graphically or algebraically during an 18-year period from 1920 to 1938, prior to the availability of digital computers. Today, the only method from that era that remains in widespread use is the McCabe-Thiele method. What are the attributes of this method that are responsible for its continuing popularity? 7.8 (a) For the cascade shown in Figure 7.39a, calculate the com- positions of streams V4 and L1. Assume atmospheric pressure, satu- rated liquid and vapor feeds, and the vapor-liquid equilibrium data given below. Compositions are in mole percent. (b) Given the feed compositions in cascade (a), how many equi- librium stages are required to produce a V4 containing 85 mol% alcohol? (c) For the cascade configuration shown in Figure 7.39b, with D= 50 mol, what are the compositions of D and L,? (d) For the configuration of cascade (b), how many equilibrium stages are required to produce a D of 50 mol% alcohol? EQUILIBRIUM DATA, MOLE-FRACTION ALCOHOL 0.1 0.2 0.3 0.5 0.7 0.9 0.5 0.68 0.82 0.94 Total condenser 100 mol 70% alcohol 30% H20 V4 4 LR D = 50 mol 4 2 100 mol 30% alcohol 70% H20 1 L1 100 mol 30% alcohol 70% H20 L1 (b) (a) Figure 7.39 Data for Exercise 7.8. 2. distillation, a number of methods were proposed for solving the equations graphically or algebraically during an 18-year period from 1920 to 1938, prior to the availability of digital computers. Today, the only method from that era that remains in widespread use is the McCabe-Thiele method. What are the attributes of this method that are responsible for its continuing popularity? 7.8 (a) For the cascade shown in Figure 7.39a, calculate the com- positions of streams V4 and L1. Assume atmospheric pressure, satu- rated liquid and vapor feeds, and the vapor-liquid equilibrium data given below. Compositions are in mole percent. (b) Given the feed compositions in cascade (a), how many equi- librium stages are required to produce a V4 containing 85 mol% alcohol? (c) For the cascade configuration shown in Figure 7.39b, with D= 50 mol, what are the compositions of D and L,? (d) For the configuration of cascade (b), how many equilibrium stages are required to produce a D of 50 mol% alcohol? EQUILIBRIUM DATA, MOLE-FRACTION ALCOHOL 0.1 0.2 0.3 0.5 0.7 0.9 0.5 0.68 0.82 0.94 Total condenser 100 mol 70% alcohol 30% H20 V4 4 LR D = 50 mol 4 2 100 mol 30% alcohol 70% H20 1 L1 100 mol 30% alcohol 70% H20 L1 (b) (a) Figure 7.39 Data for Exercise 7.8. 2.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Shunt Technology will spend $550,000 on a piece of equipment that will manufacture fine wire for the electronics industries. The shipping and installation charges will be $50,000 and net working...
-
A liquid mixture of 3 mols of water, 1 mol of propanol, 7 mols of n-butanol, 4 mols of n-pentanol, is placed in a constant pressure (1 atm) container and heated. A) At what temperature does the...
-
FAG Inc., is considering an expansion project that requires an initial fixed asset investment of $4.2 million. The fixed asset will be depreciated straight-line to zero over its three-year life,...
-
Write a structured flowchart for prob.2.3
-
Describe five characteristics of grammatically correct sentences.
-
For the case The WM. Wrigley Jr. Company: Capital Structure, Valuation, and the Cost of Capital(Darden Case: UVAF1482) please answer the following questions and explain your reasoningwhere...
-
Harold Fail owned a plot of land in Georgia known as Ebenezer Landing, where he operated a public boat ramp and parking lot. In 1999, Fail and Steve Bodiford entered into an oral agreement...
-
(Two Differences, No Beginning Deferred Taxes, Tracked through 2 Years) The following information is available for McKee Corporation for 2010. 1. Excess of tax depreciation over book depreciation ,...
-
Max Labs Inc. current portfolio of products has a 10% IRR and a 10% standard deviation. The correlation coefficients with the firm portfolio are 0.5 (r=0.5) for both, Dog Treats and Dog Food; zero...
-
An accounting entry is required, with one exception: the annual change in employee pay rates, Trans. Document ? Dec. Flowchart Description Received customer purchase order No. 53426 (Doc. No. 4) from...
-
Lena is 66 years of age, single, and blind. She is not claimed as a dependent by any other person. How much gross income must she have before she is required to file a Federal income tax return for...
-
Any restriction on commercial speech is unconstitutional. (True/False)
-
In most states, the same courts can grant both legal and equitable remedies.(True/False)
-
A law that limits the liberty of all persons to engage in a certain activity may violate constitutional provisions covering a. equal protection. b. procedural due process. c. substantive due process....
-
Bribery of public officials is strictly an ethical issue.(True/False)
-
In a civil law system, the primary source of law is a. case law. b. the decisions of administrative agencies. c. a statutory code. d. none of the above.
-
How could we avoid rushing to solutions when problem-solving? From the source: "Before you can solve a problem, you need to know what exactly you're trying to solve. Unfortunately, too many of us...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
During an economic boom, banks tend to increase their willingness to lend. How does this trend influence the actual money multiplier?
-
Using the graph below, show what happens to consumer surplus when a new technology reduces the cost of production. Po Quantity
-
Using the aggregate expenditures table below, answer the questions that follow. a. Compute the APC when income equals $2,300 and the APS when income equals $2,800. b. Compute the MPC and MPS. c. What...
-
Rank in order, from largest to smallest, the size of the friction forces in the five different situations (one or more friction forces could be zero). The box and the floor are made of the same...
-
The terminal speed of a Styrofoam ball is 15 m/s. Suppose a Styrofoam ball is shot straight down with an initial speed of 30 m/s. Which velocity graph is correct? Vy (m/s) Vy (m/s) v, (m/s) vy (m/s)...
-
Boxes P and Q are sliding to the right across a frictionless table. The hand His slowing them down. The mass of P is larger than the mass of Q. Rank in order, from largest to smallest, the horizontal...

Study smarter with the SolutionInn App



