For the following liquid junctions, determine which side of the junction will be positive: lon K*...
Fantastic news! We've Found the answer you've been seeking!
Question:
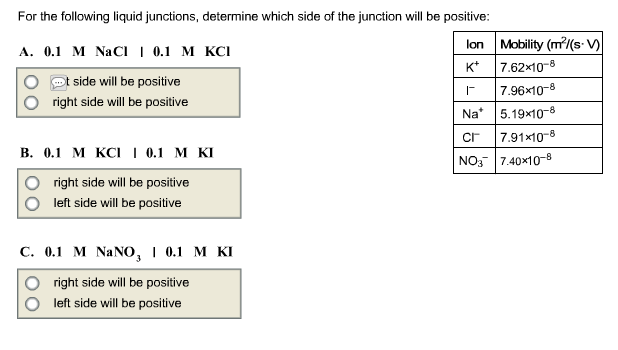
Transcribed Image Text:
For the following liquid junctions, determine which side of the junction will be positive: lon K* A. 0.1 M NaCl | 0.1 M KCI side will be positive right side will be positive B. 0.1 M KCI | 0.1 M KI right side will be positive left side will be positive C. 0.1 M NaNO, | 0.1 M KI right side will be positive left side will be positive Mobility (m²/(s.V) 7.62x10-8 | 7.96x10-8 Na 5.19x10-8 Cr 7.91x10-8 NO3 7.40×10-8 For the following liquid junctions, determine which side of the junction will be positive: lon K* A. 0.1 M NaCl | 0.1 M KCI side will be positive right side will be positive B. 0.1 M KCI | 0.1 M KI right side will be positive left side will be positive C. 0.1 M NaNO, | 0.1 M KI right side will be positive left side will be positive Mobility (m²/(s.V) 7.62x10-8 | 7.96x10-8 Na 5.19x10-8 Cr 7.91x10-8 NO3 7.40×10-8
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which side of the liquid junction 0.1 M KNO3 | 0.1 M NaCl will be negative?
-
I want to build a garage in my backyard. My neighbors driveway offers the only practical way to reach the proposed garage. I offer to purchase an easement from my neighbor, thus giving me the right...
-
I had a chance to rent my summer home for 2 weeks for $800. But I chose to have it idle. I didnt want strangers living in my summer house. What term in this chapter describes the $800? Why?
-
why people who are sophisticated but face borrowing constraint would increase consumption when they receive transfer payment?
-
Below are several transactions for Crimson Tide Corporation. A junior accountant, recently employed by the company, proposes to record the following transactions. Required: Assess whether the junior...
-
The R&D division of Jobe Corp. has just developed a chemical for sterilizing the vicious Brazilian killer bees which are invading Mexico and the southern United States. The president of Jobe is...
-
Melissa Khan alleges that on May 27, 2004, she entered into a lease and warranty agreement with Riverbank Motors Corporation, Inc. (the Dealership), for a new, 2004 Volkswagen Toureg (the Vehicle),...
-
FIFO, LIFO, and Average Cost Inventory Esplanade Company was formed on December 1, 2009. The following information is available from Esplanades inventory records for Product BAP. Units Unit Cost...
-
(a) Run linear regression on the closing price data for BTC in the range of 12/15/2020 to 12/28/2020. Report the r-squared value on your training data. Use this to predict BTC closing prices on...
-
Problem 1 Company A purchases 10,000 shares of Company B common stock for $100,000 cash. This investment represents 30% of Company B's common stock. Complete the journal entry to record the purchase...
-
2. Find the roots of the following: (10 pts. each) z6 = 2 4i b. z - 2iz - 3i 5 = 0 .
-
Blossom AG's weekly payroll of 1 9 , 7 0 0 included Social Security taxes withheld of 1 , 5 7 6 , income taxes withheld of 2 , 5 6 1 , and insurance premiums withheld of 1 9 7 . Prepare the...
-
Johnson & Jones has a cost of goods sold of $ 5 0 5 , 0 0 0 , beginning inventory of $ 1 5 0 , 0 0 0 , purchases of $ 5 0 0 , 0 0 0 , purchase returns of $ 2 5 , 0 0 0 , purchase discounts of $ 5 , 0...
-
After watching these videos; https://www.youtube.com/watch?v=2y8SA6cLUys https://youtu.be/chpCYb52_wo TASK A: Describe and detail the importance of having a well established and shared workplace...
-
Discuss the evolution of accounting theory and its significance in the contemporary business environment. Provide examples to illustrate how historical events and changing economic landscapes have...
-
A company has 4 0 0 , 0 0 0 shares with a face value of 1 . 4 euro. It announces an equity issue at 1 . 5 euros per share, with a proportion of 1 new share for 2 old shares. If shares were trading,...
-
1- A 10-year bond paying 10% coupon semi-annually is selling for $1342.61. What is the yield demanded by investors? 2- If the expected inflation rate is 6% and the real rate is 3%, what should be the...
-
As of January 1, 2018, Room Designs, Inc. had a balance of $9,900 in Cash, $3,500 in Common Stock, and $6,400 in Retained Earnings. These were the only accounts with balances in the ledger on January...
-
What would happen if charge balance did not exist in a solution? The force between two charges is given in the footnote on this page. Find the force between two beakers separated by 1.5 m of air if...
-
Explain the meaning of the quotation at the beginning of this chapter: "Get the right data. Get the data right. Keep the data right."
-
Systematic treatment of equilibrium. The acidity of Al3+ is determined by the following reactions. Write the equations needed to find the pH of Al(ClO4)3 at a formal concentration F. 2 + 2H20-Al(...
-
Study the density matrix and the partition function of a system of free particles, using the unsymmetrized wavefunction (5.4.3) instead of the symmetrized wavefunction (5.5.7). Show that, following...
-
Show that in the first approximation the partition function of a system of \(N\) noninteracting, indistinguishable particles is given by \[ Q_{N}(V, T)=\frac{1}{N ! \lambda^{3 N}} Z_{N}(V, T), \]...
-
Show that, for any law of distribution of molecular speeds, \[ \left\{\langle uangle\left\langle\frac{1}{u}ightangleight\} \geq 1 \] Check that the value of this quantity for the Maxwellian...

Study smarter with the SolutionInn App


