For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make...
Fantastic news! We've Found the answer you've been seeking!
Question:
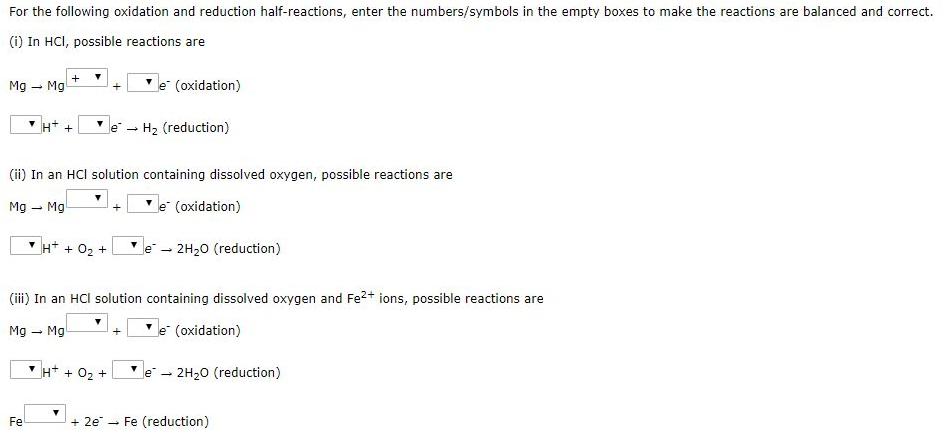
Transcribed Image Text:
For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make the reactions are balanced and correct. (i) In HCI, possible reactions are Mg - Mg e (oxidation) H* + le H2 (reduction) (ii) In an HCI solution containing dissolved oxygen, possible reactions are Mg - Mg e (oxidation) H* + 02 + 2H20 (reduction) le - (iii) In an HCI solution containing dissolved oxygen and Fe?+ ions, possible reactions are Mg – Mg e (oxidation) H+ + 02 + 2H20 (reduction) le - Fe + 2e - Fe (reduction) For the following oxidation and reduction half-reactions, enter the numbers/symbols in the empty boxes to make the reactions are balanced and correct. (i) In HCI, possible reactions are Mg - Mg e (oxidation) H* + le H2 (reduction) (ii) In an HCI solution containing dissolved oxygen, possible reactions are Mg - Mg e (oxidation) H* + 02 + 2H20 (reduction) le - (iii) In an HCI solution containing dissolved oxygen and Fe?+ ions, possible reactions are Mg – Mg e (oxidation) H+ + 02 + 2H20 (reduction) le - Fe + 2e - Fe (reduction)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
In Experiment 1, 200.0 mL of solution containing 10.0 mg of SO42- (from Na2SO4) were treated with excess BaCl2 solution to precipitate BaSO4 containing some coprecipitated Cl_. To find out how much...
-
The observations in the table are dissolved oxygen contents in water. The higher the dissolved oxygen content, the greater the ability of a river, lake, or stream to support aquatic life. In this...
-
The level of dissolved oxygen in water was measured every 2 hours in a river where industrial plants discharge processed waste. Each observation consists of four samples, from which the sample mean...
-
What are some Marketing Strategies for Delivering Objectives Polestar (Car company) has used or using?
-
Define supercoiling, positive supercoil, topoisomerase, and negative supercoil.
-
How do consumers use social networking sites when searching?
-
Consider all 40 observations on the delivery time data. Delete \(10 \%\) (4) of the observations at random. Fit a model to the remaining 36 observations, predict the four deleted values, and...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Management is concerned about the...
-
Accounting for Fair Value Hedge: Interest Rate Swap On January 1 of Year 1, Innovative Lab issued a 4-year $50,000 note to a local bank with fixed interest payments based on 6%, payable annually on...
-
Two identical particles, each having charge #q, are fixed in space and separated by a distance d. A third point charge -Q is free to move and lies initially at rest on the perpendicular bisector of...
-
And the Winner is . . . And the Number 1 for sales this month is . . . Carrie Zabrinsky smiled broadly and paused to add a little suspense to the announcement. As a development agent for Subway, she...
-
what are 2 - 3 feasible reactions to carry out in a school lab to determine their activation energy with and without a catalyst? please indicate which catalyst can be used for each and ensure there...
-
An experimental plot of In(k) vs. 1/T is obtained in lab for a reaction. The slope of the best-fit line for the graph is -1875 K. What is the value of the activation energy for the reaction in kJ/mol?
-
Please answer all correctly. I will leave an upvote. Thank you for the help. 1 . The solubility of caffeine in water is 1 g / 4 6 ml . In dichloromethane, on the other hand, 1 g can be dissolved...
-
The synthesized sample of POS is rigid, strong, and impervious to gas diffusion. Given that the amorphous density is 0.40 g/cm, which best describes the sample density? Lower than the amorphous...
-
(4) A typical long-chain fatty acid has the following chemical formula (CH3(CH2) COOH). (a) (2 pts) Identify and underline the dissociable H+. (b) (2 pts) Write the chemical formula again but in its...
-
Define the term "revenue management." Describe how an airline company could maximize revenue from available capacity at a given time.
-
Refer to the data for problem 13-36 regarding Long Beach Pharmaceutical Company. Required: Compute each division's residual income for the year under each of the following assumptions about the...
-
A student is given a crucible and asked to prove whether it is made of pure platinum. She first weighs the crucible in air and then weighs it suspended in water (density = 0.9986 g/mL). The readings...
-
Predict the value of (H f (greater than, less than, or equal to zero) for these elements at 25C (a) Br2(g); Br2 (l), (b) I2(g); I2(s)?
-
The solubility of N2 in blood at 37C and at a partial pressure of 0.80 atm is 5.6 3 1024 mol/L. A deepsea diver breathes compressed air with the partial pressure of N2 equal to 4.0 atm. Assume that...
-
W hat is diauxic growth? Explain the roles of cAMP and CAP in this process.
-
What is antisense RNA? How does it affect the translation of a complementary mRNA?
-
What are the functions of activator proteins and repressor proteins in transcription? Explain how these proteins work at the molecular level.

Study smarter with the SolutionInn App



