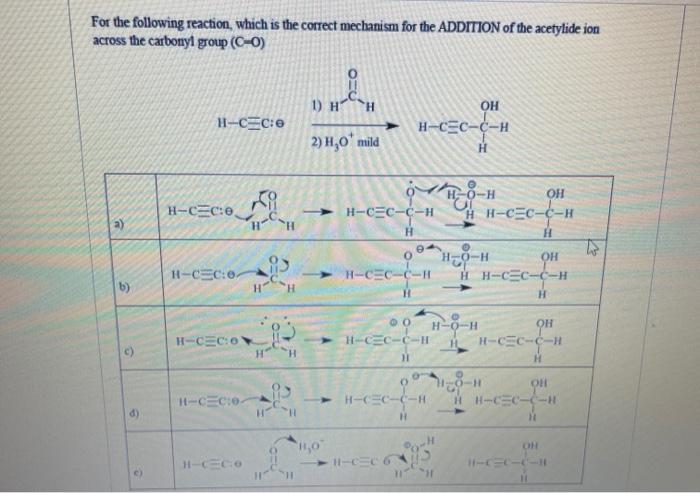
For the following reaction, which is the correct mechanism for the ADDITION of the acetylide ion...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the following reaction, which is the correct mechanism for the ADDITION of the acetylide ion across the carbonyl group (C-O) 1) H H. OH H-CEC-C-H 2) H,0' mild H0-H OH H-CEC:e H-CEC-C-H H H-CEC-C-H -> H0-H H H-CEC-C-H H-CEC:0. > H-CEC-C-H b) H. -H- H-CEC:O H-CEC-C-H H. H-CEC-C-H H-CEC:0- - H-CEC-(-H HH-CEC-C-H d) OH H-CECO H-CEC 6 H-CC-C-- For the following reaction, which is the correct mechanism for the ADDITION of the acetylide ion across the carbonyl group (C-O) 1) H H. OH H-CEC-C-H 2) H,0' mild H0-H OH H-CEC:e H-CEC-C-H H H-CEC-C-H -> H0-H H H-CEC-C-H H-CEC:0. > H-CEC-C-H b) H. -H- H-CEC:O H-CEC-C-H H. H-CEC-C-H H-CEC:0- - H-CEC-(-H HH-CEC-C-H d) OH H-CECO H-CEC 6 H-CC-C--
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
The equilibrium constant Kc equals 0.0952 for the following reaction at 227oC. What is the value of Kp at this temperature? CH3OH(g) CO(g) + 2H2(g)
-
The standard enthalpy change for the following reaction is 436.4 kJ/mol: H2(g) - H(g) + H(g) Calculate the standard enthalpy of formation of atomic hydrogen (H)?
-
What does 'buying local' mean to you? How do you determine what 'local' means? Are there limits to buying local here in PEI? Is buying local a priority for you when making purchases? How do you...
-
In what way does the auditors report on ICFR relate to managements ICFR assessment?
-
Graph several level curves of the following functions using the given window. Label at least two level curves with their z-values. z = x + y; [-4,4] [-4,4]
-
What are three ways in which you can receive income from your mutual fund investments?
-
The Fish House (TFH) in Norfolk, Virginia, sells fresh fish and seafood. TFH receives daily shipments of farm-raised trout from a nearby supplier. Each trout costs $2.45 and is sold for $3.95. To...
-
. Which describes a savings account that offers higher interest rates, but in which a * person's money must stay deposited for a specific amount of time? O CD Money Market Account Lifetime Annuity...
-
In an ideal reheat-regenerative cycle steam enters the high-pressure (H.P.) turbine at 90 bar, 600C. After expansion to 7 bar, some of the steam goes to a direct contact open heater and the balance...
-
There is much misinformation about COVID vaccines being disseminated on Facebook and other social media. Assume Congress passed a law prohibiting Facebook and other social media platforms from...
-
Question 2. Find the asymptotes, intervals of increasing-decreasing, local maximum and minimum values, point of inflection and intervals of concavity of the function y = graph. = 22-2x+5 2a: - 2 Then...
-
Find Mn to three decimal places for the definite integral, using the indicated value of n. 8 +1) dx, n = 4 (x+1) dx
-
Describe the process a company may use in screening and approving the capital expenditure budget. What is the decision rule under the net present value method? Explain with an example.
-
35 people are randomly selected and the accuracy of their wristwatches is checked, with positive errors representing watches that are ahead of the correct time and negative errors representing...
-
Define the various forms of employee empowerment and analyse the benefits of empowered employees in the service sector. Question 2 Identify the key components of the servicescape and critically...
-
Francis is trying to use influence over his coworker William. He spends endless hours at lunch talking about how he and the boss go fishing on weekends and how they both served as groomsmen in each...
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
a. Draw the resonance contributors for nitrobenzene. b. Draw the resonance contributors for chlorobenzene.
-
Suppose you are trying to synthesize the dipeptide Val-Ser. Compare the product that would be obtained if the carboxyl group of N-protected valine were activated with thionyl chloride with the...
-
In glycolysis, why must D-glucose-6-phosphate isomerize to D-fructose-6-phosphate before the cleavage reaction with aldolase occurs?
-
The annual salaries for web software development managers are normally distributed, with a mean of about \($136,000\) and a standard deviation of about \($11,500\). Random samples of 40 are drawn...
-
The scores on the SAT French Subject Test for the 2018 2020 graduating classes are normally distributed, with a mean of 603 and a standard deviation of 111. Random samples of size 16 are drawn from...
-
From 1921 through 2020, the mean return of the Standard \& Poor's 500 was \(12.59 \%\). A random sample of 38 years is selected from this population. What is the probability that the mean return for...

Study smarter with the SolutionInn App



