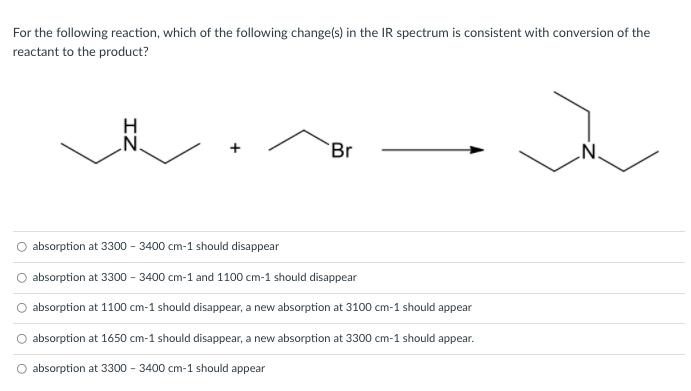
For the following reaction, which of the following change(s) in the IR spectrum is consistent with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the following reaction, which of the following change(s) in the IR spectrum is consistent with conversion of the reactant to the product? Br absorption at 3300 - 3400 cm-1 should disappear absorption at 3300 - 3400 cm-1 and 1100 cm-1 should disappear O absorption at 1100 cm-1 should disappear, a new absorption at 3100 cm-1 should appear O absorption at 1650 cm-1 should disappear, a new absorption at 3300 cm-1 should appear. O absorption at 3300 - 3400 cm-1 should appear + For the following reaction, which of the following change(s) in the IR spectrum is consistent with conversion of the reactant to the product? Br absorption at 3300 - 3400 cm-1 should disappear absorption at 3300 - 3400 cm-1 and 1100 cm-1 should disappear O absorption at 1100 cm-1 should disappear, a new absorption at 3100 cm-1 should appear O absorption at 1650 cm-1 should disappear, a new absorption at 3300 cm-1 should appear. O absorption at 3300 - 3400 cm-1 should appear +
Expert Answer:
Answer rating: 100% (QA)
IR signal of bond stretching vibrations of NH at a frequency of ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium constant for the following reaction is 1.0 Ã 10-3: Cr'. (aq) + H2EDTA2-(aq)--CrEDT A-(aq) + 2H' (aq) CH2-CO2 02GH CH EDTA N-CH,--CH2- O2C-CH2 CHy-CO2-...
-
The equilibrium constant Kc equals 0.0952 for the following reaction at 227oC. What is the value of Kp at this temperature? CH3OH(g) CO(g) + 2H2(g)
-
For the following reaction profiles, indicate The positions of reactants and products. The activation energy. ÎE for the reaction. The second reaction profile is representative of a reaction...
-
Write MATLAB code of the question. Do all parts and show code with comments and also attach a screenshot of code and outputs. Using the results of Problem 4.13, verify the following properties of WN...
-
Joe, Karen, and Larry form Gray Corporation. Joe contributes land (a capital asset) having an $8,000 adjusted basis and a $15,000 FMV to Gray in exchange for Gray ten-year notes having a $15,000 face...
-
A certain sample of uranium is reacted with fluorine to form a mixture of 235 UF6(g) and 238 UF 6 (g). After 100 diffusion steps, the gas contains 1526 235 UF 6 molecules per 1.000 10 5 total number...
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
On January 1, 2014, Peregrine Corporation acquired 100 percent of the voting stock of Osprey Corporation in exchange for $2,017,000 in cash and securities. On the acquisition date, Osprey had the...
-
Your company has decided to adopt a NoSQL database management system for storage and processing of big data. You have been asked to investigate alternative NoSQL technologies. (a) Explain the...
-
An important part of planning manufacturing capacity is having a good forecast of sales. Elizabeth Burke is interested in forecasting sales of mowers and tractors in each marketing region as well as...
-
Find the equation of the elastic curve for the simply supported beam that carries a distributed load of intensity w = wox/L. A Wo with B w = Wo L2 L FIG. P6.97
-
Bill Bluff purchased a new car from the manufacturer to sell at his automobile dealership. He purchased it using a flooring line of credit. Which entry should he make? Credit Inventory; Credit...
-
Cheryl has just completed writing the first payroll checks for her new employees. She withheld federal income tax, Social Security, and Medicare taxes from the employees' wages, and she has written a...
-
The textbook authors discuss a variety of financial ratios, and divide them into five categories. Choose three categories and calculate a financial ratios from each category for Walmart for the last...
-
Client B: What is the amount of taxable income for Elisha's self-managed super fund, Dragon Superannuation Fund, for the 2021/22 tax year? What is the final tax liability for Elisha's self-managed...
-
3. Perform analysis of the price and volume revenue drivers for each of the two products using data in Exhibit 2 . Note what you learned from the revenue driver analysis to further explain why F4S...
-
QQuirk Enterprises acquires Realcloud Inc. and records it as a business combination. Realcloud's identifiable net assets have a fair value of $30 million, and previously unrecorded intangible assets,...
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
In Section 10.11, we saw that S-adenosylmethionine (SAM) methylates the nitrogen atom of noradrenaline to form adrenaline, a more potent hormone. If SAM methylates an OH group on the benzene ring...
-
Why should 3-methylcyclohexene not be used as the starting material in Problem 30b?
-
Assume that the economy can experience four possible states: high growth, normal growth, recession, or depression. For each of those states, you expect the following stock market returns for the...
-
Suppose an investment pays off $800 or $1,600 with equal probability per $1,000 invested. What is the maximum leverage ratio you could have and still have enough to repay the loan in the event the...
-
The imposition of new trade tariffs has resulted in tensions between the United States and some of its major trading partners. Suppose you are a small business owner in the United States. a. How...

Study smarter with the SolutionInn App


