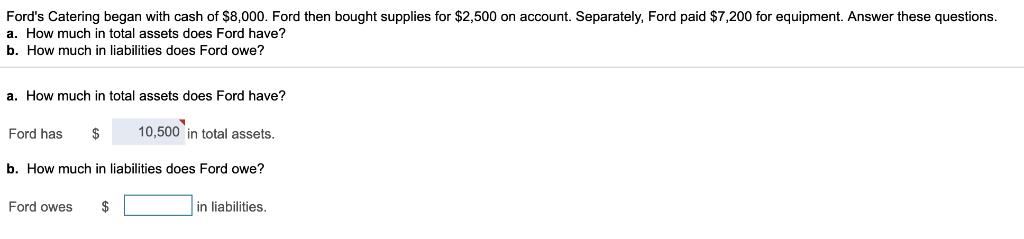
Ford's Catering began with cash of $8,000. Ford then bought supplies for $2,500 on account. Separately,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Ford's Catering began with cash of $8,000. Ford then bought supplies for $2,500 on account. Separately, Ford paid $7,200 for equipment. Answer these questions. a. How much in total assets does Ford have? b. How much in liabilities does Ford owe? a. How much in total assets does Ford have? Ford has $ 10,500 in total assets. b. How much in liabilities does Ford owe? Ford owes $ in liabilities. Ford's Catering began with cash of $8,000. Ford then bought supplies for $2,500 on account. Separately, Ford paid $7,200 for equipment. Answer these questions. a. How much in total assets does Ford have? b. How much in liabilities does Ford owe? a. How much in total assets does Ford have? Ford has $ 10,500 in total assets. b. How much in liabilities does Ford owe? Ford owes $ in liabilities.
Expert Answer:
Answer rating: 100% (QA)
Answer a Assets are the cash supplies BS and equipment fixed assets Total ... View the full answer

Related Book For 

Financial Accounting
ISBN: 978-0134725987
12th edition
Authors: C. William Thomas, Wendy M. Tietz, Walter T. Harrison Jr.
Posted Date:
Students also viewed these accounting questions
-
Fords Catering began with cash of $8,000. Ford then bought supplies for $2,500 on account. Separately, Ford paid $7,200 for equipment. Answer these questions. a. How much in total assets does Ford...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Young Software began with cash of $13,000. Young then bought supplies for $1,800 on account. Separately, Young paid $4,000 for a computer. Answer these questions. a. How much in total assets does...
-
Consider the following velocity distribution curves A and B. a. If the plots represent the velocity distribution of 1.0 L of He(g) at STP versus 1.0 L of Cl2(g) at STP, which plot corresponds to each...
-
Alert Alarm Systems installs home security systems. Two of its systems, the ICU 100 and the ICU 900, have these characteristics: The ICU 100 sells for $810 installed, and the ICU 900 sells for $1,520...
-
Use the data in Starter 5-9 to compute Pearson Education's: a. Net sales revenue b. Gross margin In Starter 5-9 Suppose Pearson Education, the publisher, sells 1,000 books on account for $150 each...
-
C. Gomez, corporate engineer of Allied Products Ltd. is proposing the replacement of existing equipment with more efficient equipment. According to information received by Gomez, the proposed...
-
Consider the ANOVA table that follows. a. Determine the standard error of estimate. About 95% of the residuals will be between what two values? b. Determine the coefficient of multiple determination....
-
Suppose you purchase a July 2020 orange juice futures contract on this day at the last price of the day. What will your profit or loss be if orange juice prices turn out to be $1.213 per pound at...
-
LaneScapes Incorporated Transactions Background LaneScapes Incorporated is a newly founded corporation in Oregon that provides premium ecologically conscious landscaping services in Lane County....
-
Briefly explain race and toggle condition with necessary logic diagram and example.
-
What are the 4 Product / Service types that can appear on your client\'s purchase and sales transactions?
-
The solubility of magnesium hydroxide, Mg(OH)2, in water is 9.0 x10^-4 g/100 mL. What volume of 0.00600 M HNO3 is required to neutralize 1.00 L of saturated Mg(OH)2 solution?
-
Potassium chloride (KCI) has a molar mass of 74.551 g/mol. Calculate the mass in grams of KCl required to prepare 50.00 mL of a 0.300 mol/L aqueous solution of KCl. Report your answer to the correct...
-
Your very smart colleague Andres Parra Munoz has been making elaborate nots for Test 2. One part of his notes reads as follows: 'Demnding domestic buyers for industry's products ssuper greater...
-
Ten grams of a sample of metal is added to 50.0 mL of water in agraduated cylinder. The final volume in the graduate is 52.8 mL.Calculate the density of the metal.
-
1 IS-LM-AD-AS: Monetary Policy Suppose the economy starts in the Full-employment equilibrium. Now, the central bank wants to achieve Stable Price. Based on a partial sticky price model by IS-LM-...
-
a. Show that the expansion of q(x) in ascending powers of x can be approximated to 10 2x + Bx 2 + Cx 3 where B and C are constants to be found. b. Find the percentage error made in using the series...
-
The Red Wagon Shop had the following inventory data: Company managers need to know the companys gross profit percentage and rate of inventory turnover for 2018 under 1. FIFO. 2. LIFO. Which method...
-
Sugar Ridge Manufacturing bought three used machines in a $167,000 lump-sum purchase. An independent appraiser valued the machines as shown: Machine No. ...................... Appraised Value 1...
-
The Juice Company reported the following comparative information at December 31, 2018, and December 31, 2017 (amounts in millions and adapted): Requirements 1. Calculate the following ratios for 2018...
-
American firms increasingly adopt stock compensation plans to compensate mid- to high-level managers. Companies are motivated to use stock options as a large portion of the compensation mix because...
-
Snappel Inc. is considering the purchase of new equipment for a purchase price of \(\$ 105,000\). The equipment has an estimated useful life of 5 years and an estimated cash salvage value of \(\$...
-
Keenlen Inc. is considering the purchase of new manufacturing equipment requiring an initial \(\$ 580,000\) investment and having an expected eight-year useful life. At the end of its life, the...

Study smarter with the SolutionInn App


