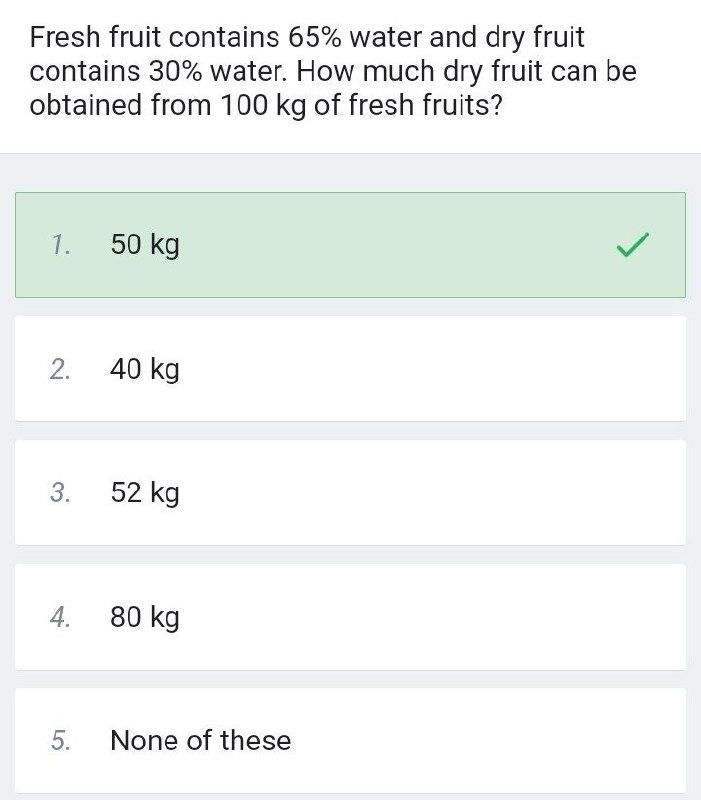
Fresh fruit contains 65% water and dry fruit contains 30% water. How much dry fruit can...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Fresh fruit contains 65% water and dry fruit contains 30% water. How much dry fruit can be obtained from 100 kg of fresh fruits? 1. 50 kg 2. 40 kg 3. 52 kg 4. 80 kg None of these 5. Fresh fruit contains 65% water and dry fruit contains 30% water. How much dry fruit can be obtained from 100 kg of fresh fruits? 1. 50 kg 2. 40 kg 3. 52 kg 4. 80 kg None of these 5.
Expert Answer:
Answer rating: 100% (QA)
Fresh fruits are of 100 kg Fresh fruit contains 65 of water Fres... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
How many alkyl halides can be obtained from monochlorination of the following alkanes? Neglect stereoisomers. (a) CH3CH2CH2CH2CH3 (b) (c) (d) (e) (f) (g) (h) (i) CH3 H3 CH CHCH2CH2CHCH3 , CH3 CH3...
-
Three arene oxides can be obtained from phenanthrene. a. Give the structures of the three phenanthrene oxides. b. What phenols can be obtained from each phenanthrene oxide? c. If a phenanthrene oxide...
-
Zinc metal can be obtained from zinc oxide, ZnO, by reaction at high temperature with carbon monoxide, CO. ZnO(s) + CO(g) Zn(s) + CO2(g) The carbon monoxide is obtained from carbon. 2C(s) + O2(g) ...
-
Timco is considering the construction of a new retail outlet. The construction cost will be 400000. Net working capital will increase by 10000. The depreciation is 10 year MACRS. The new location...
-
In the example (Limited Commitment and Market Interest Rates), suppose that v < y' and y'/y < a < b. (a) Suppose that t = t = 0. Determine the equilibrium real interest rate, and the equilibrium...
-
Write a slopeintercept equation for a line with the given characteristics. m = 2/7, y-intercept (0, -6)
-
The acentric factor can be mathematically expressed as (a) \(\omega=-1-\left.\log _{10} P_{r}^{\text {sat }} ight|_{T_{r}=0.7}\) (b) \(\omega=1-\left.\log _{10} P_{r}^{\text {sat }}...
-
Southside Corporation produces and sells a single product. Expected sales for September are 13,000 units; for October, 14,000 units; for November, 9,000 units; for December, 10,000 units; and for...
-
What strategies can organizations implement to proactively manage and resolve conflicts among employees, teams, and stakeholders, and how can organizational culture foster constructive approaches to...
-
Cherry Cotta makes custom ordered clay pots for residential gardens. Below is cost information regarding its latest job. a. Materials were purchased on account. $18,996 purchased b. A materials...
-
Kristie is a 30% partner in the KKM Partnership. During the current year, KKM reported gross receipts of $280,000 and a charitable contribution of $30,000. The partnership paid office expenses of...
-
Today one full-time American farmer feeds about people ________.
-
Calculate the value of marginal product of labor and draw the value of marginal product curve. Wandas is a fish store that hires students to pack the fish. Students can pack the following amounts of...
-
What do you believe are the major ethical challenges faced by the employees of the company in which you work? What might be done to make people in your company behave more ethically?
-
If the town owns the river and taxes cotton growers so that the efficient quantity is grown, how much tax revenue does the town receive? Is the quantity of waste zero? Explain your answer. Suppose...
-
Most of the recessions since World War II lasted _________. a) less than 6 months d) 18 to 24 months b) 6 to 12 months e) 24 to 36 months c) 12 to 18 months
-
A business entrepreneur has recently leased a small herbicide production factory. The production facility rent costs him $180 per day. In addition, he must pay $85 for the lease on conveyor line, $48...
-
(a) Use integration by parts to show that (b) If f and g are inverse functions and f' is continuous, prove that (c) In the case where f and t are positive functions and b > a > 0, draw a diagram to...
-
Show, using any necessary reagents, how the following compounds could be prepared with ethylene oxide as one of the reactants: a. CH 3 CH 2 CH 2 CH 2 OH b. CH 3 CH 2 CH 2 CH 2 D c. CH 3 CH 2 CH 2 CH...
-
Propose a mechanism for the following reaction: CHCHCf-CH2 CH CHC CHCHOCH Cl
-
a. How many alkenes could you treat with H2/Pt in order to prepare methylcyclopentane? b. Which of the alkenes is the most stable? c. Which of the alkenes has the smallest heat of hydrogenation?
-
Using the generalized virial coefficient of correlation, estimate the residual enthalpy and entropy for ethylene at \(339.7 \mathrm{~K}\) and \(1 \mathrm{bar}\), given that \(T_{\mathrm{C}}=283...
-
For a gas which obeys the equation of state \(\left(P+\frac{a}{V^{2}} ight) V=R T\), prove that the JouleThomson coefficient is \[ \mu_{\mathrm{JT}}=\frac{2 a R T}{C_{P}...
-
Show that \(\left(\frac{\partial C_{P}}{\partial P} ight)_{T}=\frac{6 B}{T^{3}}\) for a gas obeying the equation of state \(V=\frac{R T}{P}+A-\frac{B}{T^{2}}\). [Hint: We know that...

Study smarter with the SolutionInn App


