A chemical engineer is studying the rate of this reaction. 2SO3 (g) 2SO(g) + O(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
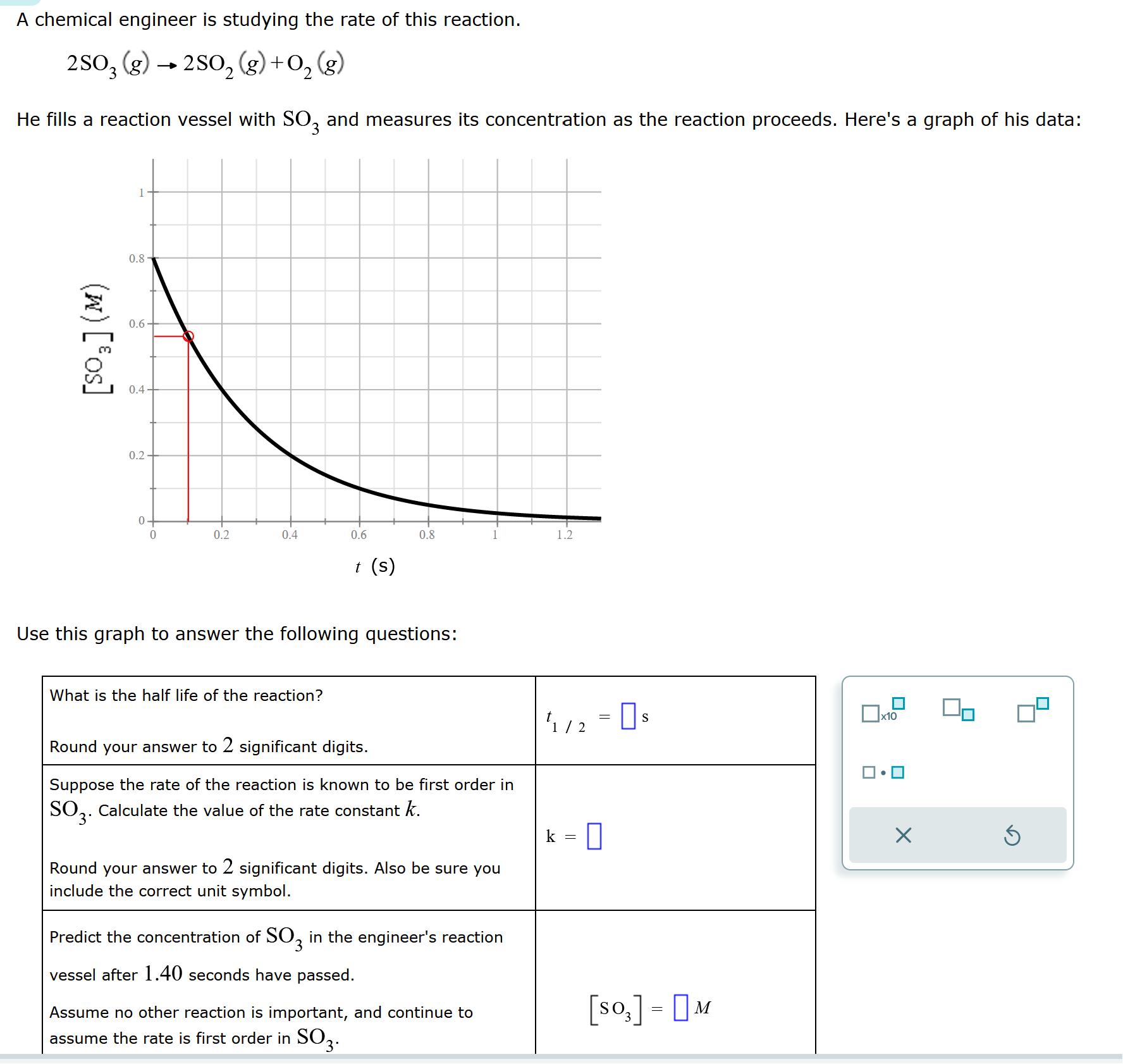
Transcribed Image Text:
A chemical engineer is studying the rate of this reaction. 2SO3 (g) → 2SO₂(g) + O₂(g) 2 He fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds. Here's a graph of his data: (w) [os] 0.8 0.6- 0.4 0.2 0+ 0 O 0.2 0.4 0.6 What is the half life of the reaction? t (s) 0.8 Use this graph to answer the following questions: Round your answer to 2 significant digits. Suppose the rate of the reaction is known to be first order in SO3. Calculate the value of the rate constant k. Round your answer to 2 significant digits. Also be sure you include the correct unit symbol. Predict the concentration of SO3 in the engineer's reaction vessel after 1.40 seconds have passed. Assume no other reaction is important, and continue to assume the rate is first order in SO3. 1.2 1/2 = 0 s k 0 [SO] - M = x10 X 5 A chemical engineer is studying the rate of this reaction. 2SO3 (g) → 2SO₂(g) + O₂(g) 2 He fills a reaction vessel with SO3 and measures its concentration as the reaction proceeds. Here's a graph of his data: (w) [os] 0.8 0.6- 0.4 0.2 0+ 0 O 0.2 0.4 0.6 What is the half life of the reaction? t (s) 0.8 Use this graph to answer the following questions: Round your answer to 2 significant digits. Suppose the rate of the reaction is known to be first order in SO3. Calculate the value of the rate constant k. Round your answer to 2 significant digits. Also be sure you include the correct unit symbol. Predict the concentration of SO3 in the engineer's reaction vessel after 1.40 seconds have passed. Assume no other reaction is important, and continue to assume the rate is first order in SO3. 1.2 1/2 = 0 s k 0 [SO] - M = x10 X 5
Expert Answer:
Answer rating: 100% (QA)
First lets define some key terms 1 Vmax is the maximum rate of an enzymecatalyzed reaction 2 kcat is ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Assume a nominal annual interest rate of i compounded semiannually. It is known that the present value today of the series of payments: X made 2n years from now and X^2 made 4n years from now is...
-
Two rats, one male and one female, scampered on board a ship anchored at a local dock. The ship set sail across the ocean. It reached a deserted island in late December. The rats abandoned the ship...
-
The Fashion Rack has a monthly accounting period. The firms chart of accounts is shown below and on the next page. The journals used to record transactions are the sales journal, purchases journal,...
-
Name the types of consumer decision-making processes. List some products you have bought using each type. Have you ever bought a product on impulse? If so, describe the circumstances.
-
On March 1, 2018, Gold Examiner receives $147,000 from a local bank and promises to deliver 100 units of certified 1-oz. gold bars on a future date. The contract states that ownership passes to the...
-
In most statistics textbooks, you will find cumulative binomial probability tables in the format shown below. These can be created using spreadsheets using the Binomial template and Data | Table...
-
A ball in motion collides elastically with an identical ball at rest. If we treat each ball as a separate system, is the work done on each ball the same?
-
Go to the Web sites of the following organizations and review their agendas and recent activities concerning assurance over sustainability disclosures: AICPA IFAC IAASB PCAOB a. How do the...
-
What is discriminating monopoly? Why a monopoly is better-off with price discrimination than without? Describe the essential conditions for price discrimination and the types of price discrimination....
-
Albert owns 100% of A Corporation, Betty is the sole proprietor of B Company, and Cai is the sole proprietor of C Company. Each business generated $500,000 of taxable income and before-tax cash flow....
-
According to Fiedler's theory of motivation, what three situational factors determine the best style of leadership for a manager to use? Describe them.
-
Which sensor uses microwave wavelengths for passive remote sensing? Explain and provide example.
-
Explain both positive and negative ways that communication has been impacted by the advancement of technology. Provide examples to support your explanation. In your opinion, does digital technology...
-
A taxpayer foreclosed on a home in TY2022 and was able to exclude $45,000 of qualified principal residence indebtedness on the basis of insolvency. However, they were not able to exclude the rest of...
-
A light spring having a spring constant of k = 125 N/m is used to pull a 9.50 kg sled on a horizontal ice rink. The coefficient of kinetic friction between the sled and the ice is M = 0.200. If the...
-
The world of online learning is another growth industry todayone that affects technical communication both directly and indirectly. Technical communicators are often, at their cores,...
-
Imagine that your friend wants to open a new hair dressing salon. She has 10 lac Taka that she wants to invest in her new venture. However, she has no knowledge of Accounting. She came to you for...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Refer to the data provided in Exercise 13-7 for Dorsey Corporation and Lansing Company. Required: Translate (remeasure) the account balances of Lansing into dollars assuming that the dollar is the...
-
In the appendix to this chapter, the Statement of Revenues, Expenditures, and Changes in Fund Balances for the General Fund for the City of Atlanta is reported. 1. How is the format used on the...
-
When should an NNO record donated services in its accounting records?
-
When your hands are cold, you can rub them together to warm them. Explain the energy transformations that make this possible.
-
A diver leaps from a high platform, speeds up as she falls, and then slows to a stop in the water. How do you define the system so that the energy changes are all transformations internal to an...
-
Figure Q10.24 shows a potential-energy diagram for a particle. The particle is at rest at point A and is then given a slight nudge to the right. Describe the subsequent motion. FIGURE Q10.24 Energy...

Study smarter with the SolutionInn App


