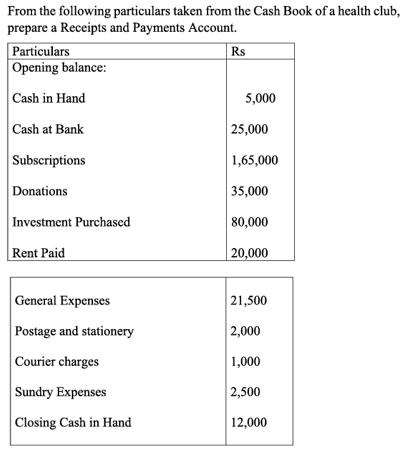
From the following particulars taken from the Cash Book of a health club, prepare a Receipts...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
From the following particulars taken from the Cash Book of a health club, prepare a Receipts and Payments Account. Particulars Opening balance: Cash in Hand Cash at Bank Subscriptions Donations Investment Purchased Rent Paid General Expenses Postage and stationery Courier charges Sundry Expenses Closing Cash in Hand Rs 5,000 25,000 1,65,000 35,000 80,000 20,000 21,500 2,000 1,000 2,500 12,000 From the following particulars taken from the Cash Book of a health club, prepare a Receipts and Payments Account. Particulars Opening balance: Cash in Hand Cash at Bank Subscriptions Donations Investment Purchased Rent Paid General Expenses Postage and stationery Courier charges Sundry Expenses Closing Cash in Hand Rs 5,000 25,000 1,65,000 35,000 80,000 20,000 21,500 2,000 1,000 2,500 12,000
Expert Answer:
Related Book For 

Accounting For Cambridge International AS And A Level
ISBN: 9780198399711
1st Edition
Authors: Jacqueline Halls Bryan, Peter Hailstone
Posted Date:
Students also viewed these accounting questions
-
From the following particulars ascertain the value of inventories as on 31st March, 2020: 3,50,000 12,00,000 18,50,000 Manufacturing Expenses *1,00,000 Selling and Distribution Expenses * 50,000...
-
The treasurer of Murray Golf Club has prepared the following receipts and payments account for the year ended 31 March 20X3: The treasurer has also supplied you with the following information: 1....
-
The treasurer of Murray Golf Club has prepared the following receipts and payments account for the year ended 31 March 20X1: The treasurer has also supplied you with the following information: 1....
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Dick Haney is opening a new business that will sell sporting goods. It will initially be a small operation, and he is concerned about the security of his assets. He will not be able to be at the...
-
Refer to the information in Problem 4-75. Required a. A local engineering firm claims it can revise the production process such that product X-2 only requires 1.6 machine hours on the shared machine....
-
The tube is 0.25 in. thick, is made of a 2014-T6 aluminum alloy and is fixed at its bottom and pinned at its top. Determine the largest axial force that it can support. 6 in. y. x P 6 in. x 10 ft P
-
3 On March 1, 2014, Rollinger Company acquired real estate on which it planned to construct a small office building. The company paid $80,000 in cash. An old warehouse on the property was razed at a...
-
An ornithologist clocks an eagle diving at some prey at 82 m/s. He knows that this particular type of eagle typically dives from a height of 57 m and can accelerate 31 m/s. How fast might the eagle...
-
Suzi Nomro operates Watercraft Supply Company, an online boat parts distributorship that is in its third year of operation. The following income statement was prepared for the year ended October 31,...
-
Corp A reports a net loss for 2 0 2 0 of $ 7 8 0 million, retained earnings at the end of the year of $ 1 7 , 8 9 3 million, and dividends during the year of $ 1 , 7 5 2 million. What was Corp A ' s...
-
You and a friend are asked to draw the two-dimensional electric field line partern for two charged objects located near each other. The charge on the first object is \(+2 q\) (with \(q\) positive),...
-
A sample of helium gas is allowed to expand in a process that is adiabatic and quasistatic. As the gas cools from \(105^{\circ} \mathrm{C}\) to \(101^{\circ} \mathrm{C}\), it does \(9.05...
-
Refer to the facts in Problem 33 and assume that the straight-line method under the alternative MACRS system was elected over the MACRS recovery period for all the assets listed. Based on these...
-
How a package is created and used in various object oriented programming paradigms?
-
Normal heptane, n C 7 H 16 n C 7 H 16 , and normal octane, n C 8 H 18 n C 8 H 18 , form ideal solutions. At 373 K 373 K , normal heptane has a vapor pressure of 106 k P a 106 k P a and normal...
-
One way to develop speaking, listening, and writing skills is to learn from others. One place to find excellent examples is through TEDTalks. For this assignment you will watch the following TEDTalk....
-
Is that Yelp review real or fake? The article A Framework for Fake Review Detection in Online Consumer Electronics Retailers (Information Processing and Management 2019: 12341244) tested five...
-
The following balances were extracted from the books of Apex Universal, a wholesaler, on 1 November 2014. Trade payables give a cash discount of 5 percent if accounts are settled within 30 days. Apex...
-
Fox Ltd has also authorized share capital of 500 000 preference shares of $0.20 each. The company decided to issue all of the preference shares at $0.45 each, as well as 8 percent debentures to the...
-
The Curtain-up Drama Club was formed several years ago. The club operates a life membership scheme. The club's treasurer is able to supply the following information about the year ended 31 December...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
The study of thermodynamics enables us to understand (a) Whether the transformation of energy is feasible or not (b) To what extent the transformation will take place (c) In which direction the...

Study smarter with the SolutionInn App



