From the following reactions at 25C: fumarate2- + NH aspartate 4,G = -36.7 kJ mol- fumarate?-...
Fantastic news! We've Found the answer you've been seeking!
Question:
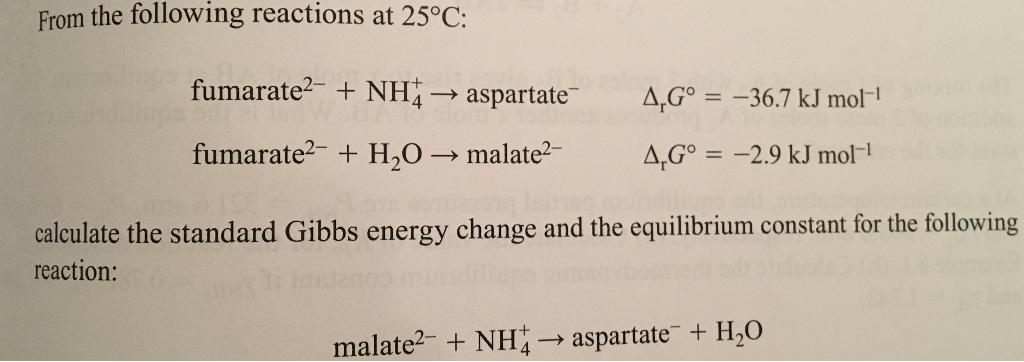
Transcribed Image Text:
From the following reactions at 25°C: fumarate2- + NH→ aspartate 4,G° = -36.7 kJ mol- fumarate?- + H,0 → malate?- 4,G° = -2.9 kJ mol- %3D calculate the standard Gibbs energy change and the equilibrium constant for the following reaction: malate2- + NH aspartate + H,O From the following reactions at 25°C: fumarate2- + NH→ aspartate 4,G° = -36.7 kJ mol- fumarate?- + H,0 → malate?- 4,G° = -2.9 kJ mol- %3D calculate the standard Gibbs energy change and the equilibrium constant for the following reaction: malate2- + NH aspartate + H,O
Expert Answer:
Answer rating: 100% (QA)
810 PM 13KBs O 33 classmate Dete Poge Standad Gibbs eneryy OG Gibbs energy of for... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Calculate the standard free energy of the following reactions at 25oC, using standard free energies of formation. a. C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(l) b. CaCO3(s) + 2H + (aq) Ca2 + (aq) + H2O(l) +...
-
Calculate the standard free energy of the following reactions at 25C, using standard free energies of formation. a. C2H4(g) + 3O2(g) 2CO2(g) + 2H2O(g) b. Na2CO3(s) + H + (aq) 2Na +(aq) + HCO3 (aq
-
Calculate the standard free-energy change and the equilibrium constant Kp for the following reaction at 25 C. See Table 18.2 for data. CO(g) 3H2(g) CH4(g) H20(g)
-
DRAW A PLAN OF YOUR HOME THEN ESTIMATE QUANTITY OF * FOOTING *BLOCK WALL UNDER DPC * BLOCK WALL ABOVE DPC * CEMENT PLASTERING OUTSIDE *GYPSUM PLASTERING INSIDE
-
Louisville Corporation produces baseball bats for kids that it sells for $32 each. At capacity, the company can produce 50,000 bats a year. The costs of producing and selling 50,000 bats are as...
-
In a random sample of males, it was found that 23 write with their left hands and 217 do not. In a random sample of females, it was found that 65 write with their left hands and 455 do not (based on...
-
Identify three functions for an ATM system.
-
On October 2, 2011, Flx, a U.S. company, entered into a forward contract to purchase 50,000 euros for delivery in 180 days at a forward rate of $0.6350. The forward contract is a derivative...
-
A lone wire connects two well-separated conducting spheres. One sphere has a radius of 10.0cm and an electric potential of 250.0V (using a reference of the zero point at infinite distance). The other...
-
Using Fig. 4.102 , design a problem that will help other students better understand Thevenin equivalent circuits. Find the Thevenin equivalent at terminals a-b of the circuit in Fig. 4.102. R3 R, R2...
-
don't answer this question According to Faraday's first law of electrolysis, the amount of any substance deposited at the electrode is directly proportional to the quantity of Select one: O a....
-
Consider the case of Greece in the spring of 2010; its national currency, the drachma, was abolished when it joined the euro-zone and it officially adopted the euro as its currency. Apply the...
-
You are the chief investment strategist for the Tiger Emerging Market Fund, which has approximately 20 percent of its assets invested in the Buenos Aires Bolsa. It is January 2001 and Argentina will...
-
The chief economist of the Lusitania country fund believes that the firm will be better served in the long term by using a composite forecasting model combining the forward rate and the Third Eye...
-
Sonys royalties payment. The U.S. sales subsidiary of the Japanese consumer electronics giant Sony Inc. is committed to paying a fixed lump sum of money (royalties) set at 2. 5 percent of its U.S....
-
Currency risk in the travel industry. Ulysses Travel Ltd (UTL) is a Boston-based travel operator that specializes in tour and holiday packages with destinations in Spain, Italy, and Greece where...
-
Eight years ago, Jacqueline de Santos purchased a brand-new condominium at the Toronto Harbourfront for $180,000. Jacqueline had been saving for her first home purchase ever since she graduated from...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
The equilibrium-constant expression for a gas reaction is Write the balanced chemical equation corresponding to this expression. Ke [C,Hg][O,]
-
A radioactive metal atom decays (goes to another kind of atom) by emitting an alpha particle (He2+ ion). The alpha particles are collected as helium gas. A sample of helium with a volume of 12.05 mL...
-
An atom in a molecule is surrounded by four pairs of electrons: one lone pair and three bonding pairs. Describe how the four electron pairs are arranged about the atom. How are any three of these...
-
A battery and four identical light bulbs are arranged in the circuit of Figure P31.3. Rank the current magnitudes at the nine lettered locations from greatest to smallest. Data from Figure P31.3 90 e...
-
A battery that initially contains \(3.0 \times 10^{24}\) electrons is used to power a light bulb for some time interval, pumping \(1.1 \times 10^{24}\) electrons through the bulb during that...
-
How can the energy conversions in an electric circuit be represented in terms of two generalized circuit elements?

Study smarter with the SolutionInn App


