Gases produced by a chemical reaction can easily be collected over water. To determine the pressure...
Fantastic news! We've Found the answer you've been seeking!
Question:

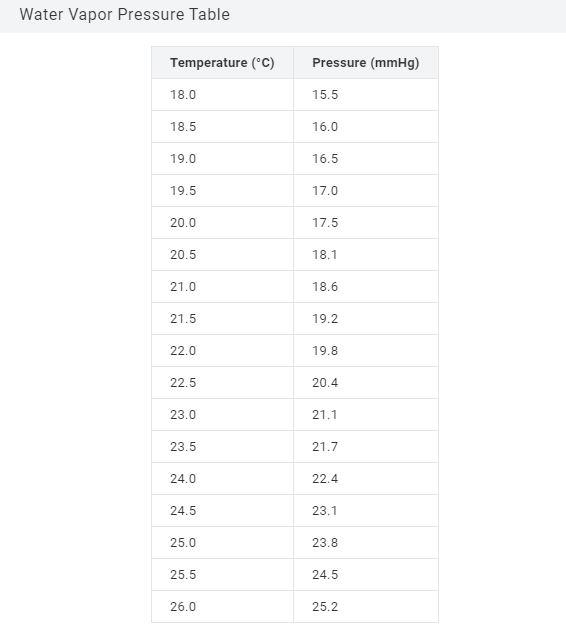
Transcribed Image Text:
Gases produced by a chemical reaction can easily be collected over water. To determine the pressure of the dry gas, the vapor pressure of the water at that temperature must be subtracted from the total pressure. 1) Consider the following reaction: Mg(s) + 2 HCl(aq) → MgCl₂(aq) + H₂(g) The total pressure of gas collected over water is 680.0 mmHg and the temperature is 22.5°C What is the pressure of hydrogen gas formed in mmHg? Pressure = mmHg Water Vapor Pressure Table Temperature (°C) 18.0 18.5 19.0 19.5 20.0 20.5 21.0 21.5 22.0 22.5 23.0 23.5 24.0 24.5 25.0 25.5 26.0 Pressure (mmHg) 15.5 16.0 16.5 17.0 17.5 18.1 18.6 19.2 19.8 20.4 21.1 21.7 22.4 23.1 23.8 24.5 25.2 Gases produced by a chemical reaction can easily be collected over water. To determine the pressure of the dry gas, the vapor pressure of the water at that temperature must be subtracted from the total pressure. 1) Consider the following reaction: Mg(s) + 2 HCl(aq) → MgCl₂(aq) + H₂(g) The total pressure of gas collected over water is 680.0 mmHg and the temperature is 22.5°C What is the pressure of hydrogen gas formed in mmHg? Pressure = mmHg Water Vapor Pressure Table Temperature (°C) 18.0 18.5 19.0 19.5 20.0 20.5 21.0 21.5 22.0 22.5 23.0 23.5 24.0 24.5 25.0 25.5 26.0 Pressure (mmHg) 15.5 16.0 16.5 17.0 17.5 18.1 18.6 19.2 19.8 20.4 21.1 21.7 22.4 23.1 23.8 24.5 25.2
Expert Answer:
Answer rating: 100% (QA)
Solution Given chemical Totul pressure of gus Temperature At table chemical reaction 5C A gus is ... View the full answer

Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these chemistry questions
-
Determine the pressure of nitrogen at 160 K, v = 0.00291 m 3 /kg using ideal gas, the van der Waals EOS, and the nitrogen table.
-
Determine the pressure of nitrogen at 160 K, v = 0.00291 m 3 /kg using the Redlich-Kwong EOS and the nitrogen table.
-
Determine the pressure of nitrogen at 160 K, v = 0.00291 m 3 /kg using the Soave EOS and the nitrogen table.
-
Consider the following (unoptimized) MIPS assembler program: 2 3 4 5 6 6 - 7 BA 8 0 9 10 10 .text .globl main #int procB (int a, int b) # { # # # # # # } int = 0; c = a + b return C; 44 11 procB: 12...
-
How is risk aversion reflected in the SML?
-
How can organizations foster a culture of diversity, equity, and inclusion within teams, proactively addressing systemic biases and promoting equitable access to opportunities for professional growth...
-
In Exercises 1 to 4, it may be helpful to draw a figure such as Figure 5.5. Figure 5.5. Using the normal curve table, determine the area of the standard normal distribution that is less than the...
-
Seattle Roast Coffee Company produces Columbian coffee in batches of 8,000 pounds. The standard quantity of materials required in the process is 8,000 pounds, which cost $5.00 per pound. Columbian...
-
Why is Microsoft Corp bond a good investment? The Bond has a maturity date of 6/1/2039 and a coupon rate of 5.2000% and is At the current price of 114.234 USD, this equates to a 4.16 percent annual...
-
Case Introduction Established in 1967, Denns Fish House is a regional restaurant headquartered in a Southern US City, with dine-in buffet restaurants and several fast-food franchise locations....
-
In an IT company, a project manager needs to assign four projects to four different and independent teams. Any team can handle only one project and vice-versa. Based on his past experience, he has...
-
Assume a Hotelling line of a distance l = 1. There are two companies A and B, both of them being located at a longer distance. Company A is placed closer to the end of the line, while company B is...
-
4. Asian women in the US are more likely to study in college and more likely to study a science major than Latino women. a. Science majors have many more male students than female students. If most...
-
This is an Industrial Organization Economics Question: Suppose that demand for rollerblades is given by D(p) = A ? p. The cost function for all firms is C(y) = wy2 + f , where f is a fixed set-up...
-
Suppose you are stranded on an island. Luckily, this island is rich in clams and mangos. If you devote all of your time to harvesting clams, you can get 100 clams in a week. If you us all of your...
-
Year Quarter Units 1 Q1 20 1 Q2 100 1 Q3 175 1 Q4 13 2 Q1 37 2 Q2 136 2 Q3 245 2 Q4 26 3 Q1 75 3 Q2 155 3 Q3 326 3 Q4 48 4 Q1 92 4 Q2 202 4 Q3 384 4 Q4 82 5 Q1 176 5 Q2 282 5 Q3 445 5 Q4 181 Using...
-
What is the after-tax cash flow for the sale of this asset? Initial cost is $500,000 depreciated using MACRS 5-year recovery Sold after 3 years for $200,000 The tax rate is 28%
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Air in a typical house is completely exchanged with outside air about every two hours unless care is taken to seal cracks. If the outside air is at 20 8F with 80% humidity and the inside air is...
-
The 4 kg of saturated liquid water in Fig. 2.30 are completely vaporized at a constant pressure of 200 kPa. Determine the volume of the water at state 1 and at state 2. Figure 2.30 State 1 State 2...
-
Refrigerant 134a undergoes a process from 400 kPa and 400 C to a pressure of a) 400 kPa, b) 800 kPa, c) 1200 kPa in a rigid container. Calculate the heat transfer and the change in specific entropy.
-
Calculate the volatility and the current price of oil futures implied by the call 1600 August and the call 1700 August of Figure 19.8 by using the Black-Scholes formula with \(T=.25\). OIL CRUDE OIL...
-
A company issues a \(10 \%\) coupon bond that matures in 5 years. However, this company is in trouble, and it is estimated that each year there is a probability of .1 that it will default that year....
-
Consider a continuous-time environment, with \(e\) as a variable outside of the market. (a) Suppose the final payoff is \(V\left(x_{e}, T ight)=x_{e}(T)\). Find \(V\left(x_{e}, t ight)\). (b) Find a...

Study smarter with the SolutionInn App


