?Given these reactions,what is ?Hrxn for this reaction? Given these reactions, X(s) +-0(g) XO(S) XCO3(s)
Fantastic news! We've Found the answer you've been seeking!
Question:
?Given these reactions,what is ?Hrxn for this reaction?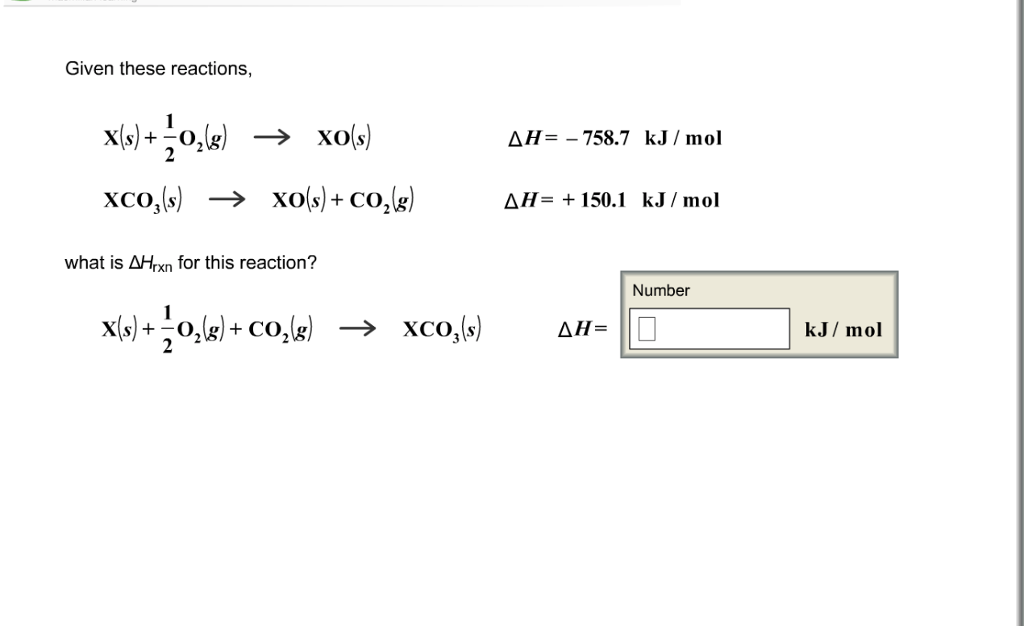
Transcribed Image Text:
Given these reactions, X(s) +-0₂(g) → XO(S) XCO3(s) → XO(s) + CO₂(g) what is AHrxn for this reaction? X(s) + O₂(g) + CO₂(g) → XCO3(s) AH-758.7 kJ/mol AH= + 150.1 kJ/mol ΔΗ= Number 0 kJ/ mol Given these reactions, X(s) +-0₂(g) → XO(S) XCO3(s) → XO(s) + CO₂(g) what is AHrxn for this reaction? X(s) + O₂(g) + CO₂(g) → XCO3(s) AH-758.7 kJ/mol AH= + 150.1 kJ/mol ΔΗ= Number 0 kJ/ mol
Expert Answer:
Answer rating: 100% (QA)
Given X s 0 9 2 XC03 5 Find A texn for this XO S Solution ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
For the complete redox reactions given here, (i) break down each reaction into its half-reactions; (ii) identify the oxidizing agent; (iii) identify the reducing agent. (a) (b) (c) (d) 2Sr + O, 2SrO
-
For the complete redox reactions given here, write the half-reactions and identify the oxidizing and reducing agents: (a) (b) (c) (d) Cl2 + 2NaBr_ 2NaCl + Br- Si + 2F,-> SF
-
Given this reaction at equilibrium: CO(g) + Br2(g) + COBr2(g) In which directiontoward reactants or toward productsdoes the reaction shift if the equilibrium is stressed by each change? 1. Br2 is...
-
Refer to all of the facts in Problem 12-6. Required 1. Using the format in the chapters appendix, prepare a statement of cash flows work sheet. 2. Prepare a statement of cash flows for 2014 using the...
-
A recent annual report for FedEx contained the following data: Required: 1. Determine the receivables turnover ratio and average days sales in receivables for the current year. 2. Explain the meaning...
-
Criticize the following quotation: We shall have no difficulty in paying for new plant assets needed during the coming year because our estimated outlays for new equipment amount to only $80,000, and...
-
Plaintiff grounds manager sued a manufacturer, Monsanto, alleging that herbicide use caused his non-Hodgkins lymphoma. The jury awarded the plaintiff \($39.3\) million in compensatory damages and...
-
The Metropolitan Bus Company (MBC) purchases diesel fuel from American Petroleum Supply. In addition to the fuel cost, American Petroleum Supply charges MBC $250 per order to cover the expenses of...
-
List and describe/discuss the five advantages of intermediate sanctions over traditional probation or incarceration.
-
W&S Partners has just won the January 31, 2026, audit for Cloud 9. As a part of the risk assessment phase for the new audit, the audit team needs to gain an understanding of Cloud 9's structure and...
-
5 years ago, the age of Diksha's grandfather was 6 times that of Diksha and after 10 years from now, her Grandfather will be 3 times of her age. Find the ratio of Diksha and her Grandfather's present...
-
Textflix is a new idea to help reduce college student cost of textbooks. Textflix is a new project to be undertaken by the Netflix Corporation. Textflix will charge $20 per month to have access to a...
-
(a) A solid solution is obtained by adding CaO to Ta205. Write two defect equations to describe the defect chemistry that results. (b) Based on the defect models you wrote in part (a) and using the...
-
November 1, 2023 Dear Sir: We have reviewed CLL's internal third-quarter financial statements, dated August 31, 2023. As a result of this review, we have determined that your financial raos connue to...
-
Luna wants enough life insurance to cover $30,000 per year until her 8-year-old turns 18. Using the Capital Drawdown method, what life insurance is recommended to meet Luna's needs?
-
In this problem, we want to determine the Fourier series of the steady state output y(t), of a Butterworth filter given by H(s) below, to the input x(t). The input x(t) is a periodic rectangular...
-
Consider f: C\{0} C by (2) == / (a) Writing z = x+iy find the real and imaginary u: R\{(0,0)} R and v R\{(0,0} R parts of f. (b) Compute the partial derivatives: du du dv v and 1 ' (c) Compute...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
You may need Table to answer the following questions. a. Which is the stronger acid, HCl or H2O? b. Which is the stronger acid, H2O or HNO2? c. Which is the stronger acid, HCN or HOC6H5? Table...
-
Molybdenum is obtained as a by-product of copper mining or is mined directly (primary deposits are in the Rocky Mountains in Colorado). In both cases it is obtained as MoS2, which is then converted...
-
The radioactive isotope 247Bk decays by a series of or-particle and -particle productions, taking 247Bk through many transformations to end up as 207Pb. In the complete decay series, how many a...
-
The global market presents firms with more complex ethical issues than they would experience if operations were limited to one country and one culture. Moral standards vary across cultures. In some...
-
Bhopal is a city in central India with a population, in 1984 , of 800,000 . Because it was, at that time, home to the largest mosque in India, Bhopal was a major railway junction. Its main industries...
-
PricewaterhouseCoopers (or PwC, as it is known), one of the United States' "Big 4" accounting firms, has had a tax practice in Russia since the time that country changed from Communist rule. One of...

Study smarter with the SolutionInn App


