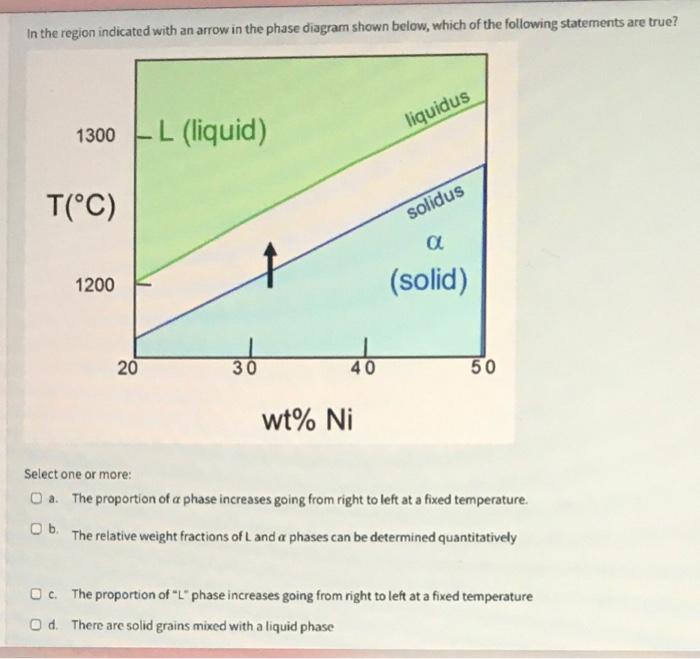
In the region indicated with an arrow in the phase diagram shown below, which of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
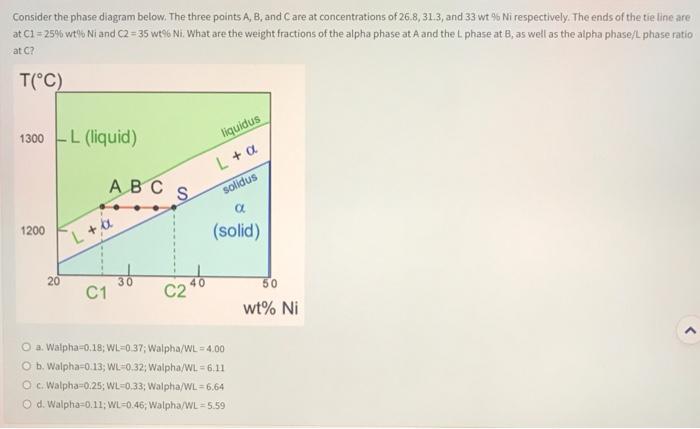
In the region indicated with an arrow in the phase diagram shown below, which of the following statements are true? 1300 L (liquid) - . T(°C) 1200 20 36 40 wt% Ni liquidus solidus α (solid) 50 Select one or more: a. The proportion of a phase increases going from right to left at a fixed temperature. b. The relative weight fractions of L and a phases can be determined quantitatively c. The proportion of "L" phase increases going from right to left at a fixed temperature Od. There are solid grains mixed with a liquid phase Consider the phase diagram below. The three points A, B, and C are at concentrations of 26.8, 31.3, and 33 wt% Ni respectively. The ends of the the line are at C1 = 25% wt% Ni and C2 = 35 wt% Ni. What are the weight fractions of the alpha phase at A and the L phase at B, as well as the alpha phase/L phase ratio at C? T(°C) 1300-L (liquid) 1200 20 ABC L+a 30 C1 liquidus L + a solidus a (solid) S C2 O a. Walpha 0.18; WL-0.37; Walpha/WL=4.00 O b. Walpha-0.13; WL 0.32; Walpha/WL=6.11. O c. Walpha 0.25; WL-0.33; Walpha/WL = 6.64 O d. Walpha=0.11; WL-0.46, Walpha/WL-5.59 50 wt% Ni In the region indicated with an arrow in the phase diagram shown below, which of the following statements are true? 1300 L (liquid) - . T(°C) 1200 20 36 40 wt% Ni liquidus solidus α (solid) 50 Select one or more: a. The proportion of a phase increases going from right to left at a fixed temperature. b. The relative weight fractions of L and a phases can be determined quantitatively c. The proportion of "L" phase increases going from right to left at a fixed temperature Od. There are solid grains mixed with a liquid phase Consider the phase diagram below. The three points A, B, and C are at concentrations of 26.8, 31.3, and 33 wt% Ni respectively. The ends of the the line are at C1 = 25% wt% Ni and C2 = 35 wt% Ni. What are the weight fractions of the alpha phase at A and the L phase at B, as well as the alpha phase/L phase ratio at C? T(°C) 1300-L (liquid) 1200 20 ABC L+a 30 C1 liquidus L + a solidus a (solid) S C2 O a. Walpha 0.18; WL-0.37; Walpha/WL=4.00 O b. Walpha-0.13; WL 0.32; Walpha/WL=6.11. O c. Walpha 0.25; WL-0.33; Walpha/WL = 6.64 O d. Walpha=0.11; WL-0.46, Walpha/WL-5.59 50 wt% Ni
Expert Answer:
Answer rating: 100% (QA)
The given phase diagram having separation for liquid and solid The correct statem... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Which of the following statements are true regarding dividends? _____ 1. Cash and stock dividends reduce retained earnings. _____ 2. Dividends payable is recorded at the time a cash dividend is...
-
Which of the following statements are true of long-term investments? a. They can include investments in trading securities. b. They are always easily sold and therefore qualify as being marketable....
-
Which of the following statements are true for discrete random variables? Explain why each statement is true or false. a. The mean can be negative. b. The mean can be zero. c. The standard deviation...
-
A business student has $3,500 available from a summer job and has identified three potential stocks in which to invest. The cost per share and expected return over the next two years are given in the...
-
The chapter describes how a batch processing system works with a used book shop as an example. Looking at Figure 3.2 and its description of how the system works, identify another type of business...
-
It is common to use the normalization \(u^{(\alpha) \dagger}(\boldsymbol{p}) u^{(\beta)}(\boldsymbol{p})=(E / m) \delta_{\alpha \beta}\) for a massive free Dirac spinor \(u^{(\alpha)}\), where \(E\)...
-
Should the requirements of the UCC be subject to the application of reliance theories? Go back and review the facts in Case 21-3 about the coal contract. Should silence followed by contract execution...
-
Condensed financial data of Sinjh Inc. follow. Additional information: 1. New plant assets costing $80,000 were purchased for cash during the year. 2. Old plant assets having an original cost of...
-
What is Online transaction processing and Online Analytical Processing? How are they different?
-
Kofi Allon, who is 32 years old and single, is employed as a technical consultant for a large electronics distributor. He lives at 678 Birch Street, LaMesa, CA 91941. Kofi's Social Security number is...
-
If 1=3 2=3 3=5 4=4 5=4 Then, 6=? Please share the correct answer
-
Sankey diagrams for various countries are available at http://flowcharts.llnl.gov. Obtain the flowcharts for China, Germany, Saudi Arabia, and USA for 2011. Compare the overall efficiency of each...
-
Briefly explain whether you agree with the following statement: Japan has always been much more heavily involved in international trade than are most other nations. In fact, today Japan exports a...
-
Why did Congress decide to establish the Federal Reserve System in 1913?
-
What is stagflation? Why might a supply shock result in it?
-
Suppose that you are the owner of a professional baseball team in a major city, and MLB allows a second team to locate in your city. Describe and show using a graph the potential impact on your...
-
Consider the Initial Value Problem consisting of the ODE and the Initial Condition given below. dy dt y(t=0) = 2 a) State whether the ODE is linear or nonlinear. b) Express the ODE as an algebraic...
-
The slopes of the tangents at the points where the curve y = x2 - 4x intersects the X-axis is 1) 1 2) +2 3) +3 4) +4
-
Characterize each of the following atoms as being either nucleophilic or electrophilic. (a) Iodide ion, I- (b) Hydrogen ion, H+ (c) Carbon in methyl cation, +CH3 (d) Sulfur in hydrogen sulfide. H2S...
-
The structure of the terpene limonene is shown in the margin. Identify the two 2-methyl-1,3-butadiene (isoprene) units in limonene. (a) Treatment of isoprene with catalytic amounts of acid leads to a...
-
When 1-pentene is treated with mercuric acetate, followed by sodium borohydride, which of the following compounds is the resulting product? (a) 1-Pentyne; (b) pentane; (c) 1-pentanol; (d) 2-pentanol.
-
Please reflect on and explain the role and usefulness of the concept of SD in relation to the protection of the environment.
-
Has the concept of SD achieved the balance between all three pillars: environmental protection; economic development; and social issues?
-
What, if any, is the normative content of the concept of SD?

Study smarter with the SolutionInn App


