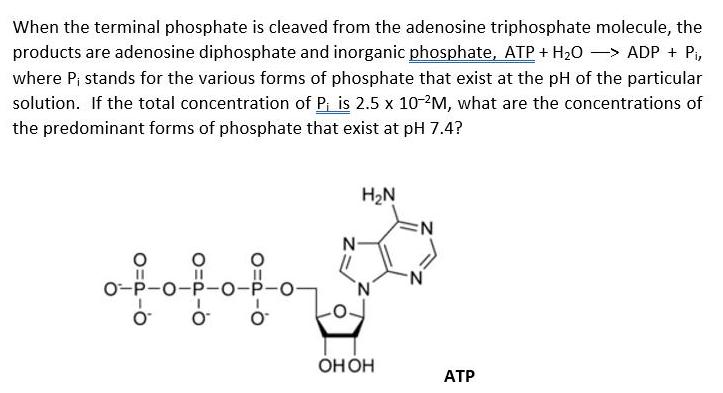
When the terminal phosphate is cleaved from the adenosine triphosphate molecule, the products are adenosine diphosphate...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
When the terminal phosphate is cleaved from the adenosine triphosphate molecule, the products are adenosine diphosphate and inorganic phosphate, ATP + H20 –> ADP + P, where P; stands for the various forms of phosphate that exist at the pH of the particular solution. If the total concentration of P, is 2.5 x 10-?M, what are the concentrations of the predominant forms of phosphate that exist at pH 7.4? H2N N: N- 0-P-0-P-O-P-O- Онон АТР When the terminal phosphate is cleaved from the adenosine triphosphate molecule, the products are adenosine diphosphate and inorganic phosphate, ATP + H20 –> ADP + P, where P; stands for the various forms of phosphate that exist at the pH of the particular solution. If the total concentration of P, is 2.5 x 10-?M, what are the concentrations of the predominant forms of phosphate that exist at pH 7.4? H2N N: N- 0-P-0-P-O-P-O- Онон АТР
Expert Answer:
Answer rating: 100% (QA)
In serum phosphate exists in two forms dihydrogen phosphate H2PO4 and its salt monohydro... View the full answer

Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony-Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these chemistry questions
-
What are the usual ionic forms of ATP and ADP in typical cells? Does this information have any bearing on the free-energy change for the conversion of ATP to ADP?
-
Phosphate buffers are important in regulating the pH of intracellular fluids at pH values generally between 7.1 and 7.2. a. What is the concentration ratio of H2PO4- to HPO4-2 in intracellular fluid...
-
When a researcher reports a p value, p stands for probability or risk. a. What probability does this p refer to? b. Do we typically want p to be large or small? c. What is the conventional standard...
-
Use the method of maximum likelihood to estimate in the pdf f Y (y; ) = /2y e y , y 0 Evaluate e for the following random sample of size 4: Y 1 = 6.2, Y 2 = 7.0, Y 3 = 2.5, and Y 4 = 4.2.
-
What is adverse selection? How do insurers try to prevent adverse selection?
-
Give an example of a healthcare product that is financed by the government but produced by private firms. Can you explain why this arrangement exists?
-
What are opportunity costs? Why are they important to managers? What is the opportunity cost of not investing in environmental protection? Provide an example to support your answer.
-
The Brick Company had cash sales of $280,000 for 2018, its first year of operation. On April 2, the company purchased 210 units of inventory at $390 per unit. On September 1, an additional 160 units...
-
Whether job/career changes occur because of automation, pandemic, political turmoil, global competition, or other technology innovations, what specific and/or general job skills do you think will be...
-
Hook Industries is considering the replacement of one of its old drill presses. Three alternative replacement presses are under consideration. The relevant cash flows associated with each are shown...
-
Refer to the mortgage data below, observed on Feb 16, 2018, and provide a detailed explanation for each of the pair-wise mortgage rate differences for these three mortgages. Interest rate 5.14%5...
-
Consider a model with two periodsthe first time period is the four years after high school and the second time period is the next 40 years. A person without a college education receives \($120,000\)...
-
A research group claims that the median age of the users of a social networking website is less than 32 years old. In a random sample of 20 users, 5 are less than 32 years old, 13 are more than 32...
-
A financial services accountant claims that the median amount of financial debt for families holding such debts is less than \(\$ 60,000\). In a random sample of 70 families with financial debt, the...
-
In determining the primary responsibility of the external auditor for a company's financial statements, the auditor owes primary allegiance to: a. The management of the company being audited because...
-
During a weather report, a meteorologist claims that the median daily high temperature for the month of January in San Diego is \(66^{\circ}\) Fahrenheit. The high temperatures (in degrees...
-
Brooklyn is going to finance new office equipment at a 1% rate over a 2.5 year term. If she can afford monthly payments of $125, how much can she pay for the new office equipment? Round your answer...
-
1A. If the researcher is concerned about the number of variables, the nature of the analysis, and completion rates, then, he/she is at which stage of the sampling design process (Figure 11.1 in the...
-
Te oxidation of glucose to CO2 and water is a major source of energy in aerobic organisms. It is a reaction favored mainly by a large negative en-thalpy change. a. At 37C, what is the value for Go?...
-
Suppliers of radioisotopically labeled compounds usually provide each product as a mixture of labeled and unlabeled material. Unlabeled material is added deliberately as a carrier, partly because the...
-
Based on information presented on pages 402-403, sketch curves relating glycogen synthase reaction velocity to [UDP-glucose], for both the a and b forms of the enzyme, in the presence and absence of...
-
Yvonne had the following transactions in the shares of Scotia plc: Sally's capital gains tax position in 2023-24 was as follows: You are required to: (a) Calculate Yvonne's chargeable gain. (b)...
-
In February 2014, Maria bought a diamond necklace for 13,500. In 2017 the necklace was stolen and, as a result, an insurance payment of 14,000 was received in February 2018. In the same month, Maria...
-
Jerome made the following gifts to family members during tax year 2023-24: (1) On 28 May 2023, Jerome made a gift of a house valued at 187,000 to his wife. His uncle had originally purchased the...

Study smarter with the SolutionInn App


