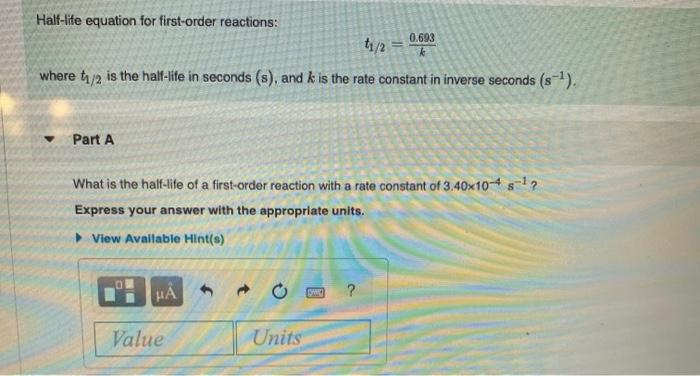
Half-life equation for first-order reactions: t/2 = where t/2 is the half-life in seconds (s), and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
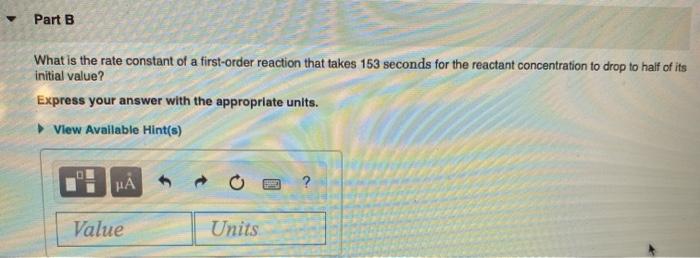
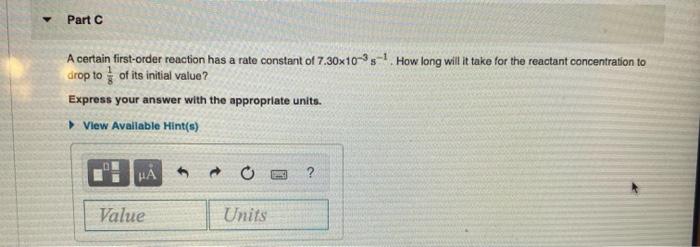
Half-life equation for first-order reactions: t₁/2 = where t₁/2 is the half-life in seconds (s), and k is the rate constant in inverse seconds (s-¹). Part A What is the half-life of a first-order reaction with a rate constant of 3.40x104s¹? Express your answer with the appropriate units. View Available Hint(s) HÅ Value Units 0.693 (PHIC) ? Part B What is the rate constant of a first-order reaction that takes 153 seconds for the reactant concentration to drop to half of its initial value? Express your answer with the appropriate units. View Available Hint(s) μà Value Units Part C A certain first-order reaction has a rate constant of 7.30x10-3s¹ How long will it take for the reactant concentration to drop to of its initial value? Express your answer with the appropriate units. ▸ View Available Hint(s) HA Value 0 Units Half-life equation for first-order reactions: t₁/2 = where t₁/2 is the half-life in seconds (s), and k is the rate constant in inverse seconds (s-¹). Part A What is the half-life of a first-order reaction with a rate constant of 3.40x104s¹? Express your answer with the appropriate units. View Available Hint(s) HÅ Value Units 0.693 (PHIC) ? Part B What is the rate constant of a first-order reaction that takes 153 seconds for the reactant concentration to drop to half of its initial value? Express your answer with the appropriate units. View Available Hint(s) μà Value Units Part C A certain first-order reaction has a rate constant of 7.30x10-3s¹ How long will it take for the reactant concentration to drop to of its initial value? Express your answer with the appropriate units. ▸ View Available Hint(s) HA Value 0 Units
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below ... View the full answer

Related Book For 

Discovering Advanced Algebra An Investigative Approach
ISBN: 978-1559539845
1st edition
Authors: Jerald Murdock, Ellen Kamischke, Eric Kamischke
Posted Date:
Students also viewed these chemistry questions
-
How long will it take for monthly payments interest rate on the loan is: a. 6% compounded monthly? b. 7% compounded monthly? c. 8% compounded monthly? d. 9% compounded monthly
-
How long will it take for monthly payments interest rate on the loan is: a. 7.5% compounded annually? b. 7.5% compounded semiannually? c. 7.5% compounded quarterly? d. 7.5% compounded monthly?
-
How long will it take a 1750-W motor to lift a 315-kg piano to a sixth-story window 16.0m above?
-
A Moving to another question will save this response. Question 9 1 L The absent of the logic action Release in the Resource Module means: The operator will rework the entity a. b. C. The operator...
-
Why are money market securities sometimes referred to as cash equivalents?
-
a. With random sampling, does the shape of the data distribution tend to resemble more closely the sampling distribution or the population distribution? Explain. b. Is the sampling distribution of...
-
Many state courts have forms posted on their websites. Determine whether California has a form for notice of removal of actions for federal court.
-
Nick Company had the following transactions during April. 1. Sold merchandise on account. 2. Purchased merchandise on account. 3. Collected cash from a sale to Athletic Company. 4. Recorded accrued...
-
Describe the change method/theory you feel would be the most effective in a large organization, and why. Describe how you would create an intrapreneurship mindset within a larger organization....
-
FIT ($1,200.58 - $221.15 = $979.43 taxable)...................... (86.00)* how did you get the answer 86
-
During the past year, Gutter Corp., operated within the relevant range of its fixed costs. Monthly production volume during the year ranged from 60,000 to 100,000 units of product and corresponding...
-
Pronghorn Design Works generated $315,840 in operating income on sales revenue of $2,632,000. The company had $1,860,000 in assets on January 1 and $2,840,000 in assets on December 31. (a-b) (a)...
-
The following items are necessary for preparing the 2 0 2 4 year - end adjustments for Rogers Advertising Agency. Rogers s fiscal year - end is December 3 1 . On July 1 , 2 0 2 4 , Rogers received $...
-
ABC Advertising, owned by Mr Anish, provides advertising consulting services. During January 2 0 2 1 , the following events occurred: Jan. 2 Owner contributed Rs . 5 0 , 0 0 0 and a new computer...
-
Heart Lake developments sold four lakefront lots for $ 3 0 , 5 0 0 per hectare. If the sizes of the lots in hectares were 3 1 2 , 2 3 7 , 5 5 8 , and 4 4 9 respectively, what was the total sales...
-
Jeff Bezos is opening a Amazon fulfillment center in South Park, Colorado, and employs Stephen Stotch for q hours a day for a wage of p. Jeff's profits are = V(q)- p, where V(g), is increasing and...
-
People always work more efficiently in large organizations. True False
-
What are three disadvantages of using the direct write-off method?
-
Decide whether each expression is an identity by substituting values for A and B. a. cos (A + B) = cos A + cos B b. sin (A + B) = sin A + sin B c. cos (2A) = 2 cos A d. sin (2A) = 2 sin A
-
Create a data set of 9 values such that the median is 28, the minimum is 11, and there is no upper whisker on a box plot of the data.
-
Los Angeles, California, and Honolulu, Hawaii, are about 2500 mi apart. One plane flies from Los Angeles to Honolulu, and a second plane flies in the opposite direction. a. Describe the meaning of...
-
Can you explain why the annual report continues to be a useful document?
-
Can you explain why analysts might view conference calls/investor presentations as more important than the annual report?
-
How would the budget-setting philosophy of a service provider differ from that of a manufacturing business?

Study smarter with the SolutionInn App


