Harrison Company reports the following cost information for August: Cost of goods manufactured Finished goods inventory,...
Fantastic news! We've Found the answer you've been seeking!
Question:
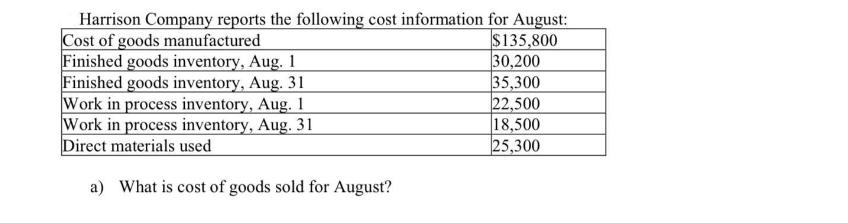

Transcribed Image Text:
Harrison Company reports the following cost information for August: Cost of goods manufactured Finished goods inventory, Aug. 1 Finished goods inventory, Aug. 31 Work in process inventory, Aug. 1 Work in process inventory, Aug. 31 Direct materials used a) What is cost of goods sold for August? $135,800 30,200 35,300 22,500 18,500 25,300 b) What is the amount of direct labor incurred by Harrison Company in August? c) What is the amount of direct materials used by Harrison Company in August? d) What is the amount of manufacturing overhead incurred by Harrison Company in August? Harrison Company reports the following cost information for August: Cost of goods manufactured Finished goods inventory, Aug. 1 Finished goods inventory, Aug. 31 Work in process inventory, Aug. 1 Work in process inventory, Aug. 31 Direct materials used a) What is cost of goods sold for August? $135,800 30,200 35,300 22,500 18,500 25,300 b) What is the amount of direct labor incurred by Harrison Company in August? c) What is the amount of direct materials used by Harrison Company in August? d) What is the amount of manufacturing overhead incurred by Harrison Company in August?
Expert Answer:
Answer rating: 100% (QA)
a To calculate the cost of goods sold COGS for August we need to consider the change in finish... View the full answer

Related Book For 

Financial and Managerial Accounting Using Excel for Success
ISBN: 978-1111993979
1st edition
Authors: James Reeve, Carl S. Warren, Jonathan Duchac
Posted Date:
Students also viewed these accounting questions
-
Match the expression in Column I with its equivalent expression in Column II. Choices may be used once, more than once, or not at all. (a) (b) (c) (d) 2 5 I -2 2 5 (3) -(--13) 2 5 7 A. B. C. D. II...
-
Computing COGM and COGS Acronyms: The following cost information are gathered from Drusilla Inc. for the month ended June 30: BWIP Beginning Work-in-process inventory Costs of direct materials used...
-
The Cost Department of Columbus Company received the following monthly data, pertaining solely to manufacturing activities, from the general ledger clerk: Additional data: (a) Physical inventory...
-
You have learned a great deal about the Internet Protocol (IP). IP is a set of rules for how data is sent across networks and arrive at the intended destination. An IP address is a numeric identifier...
-
A thin-walled, hollow spherical shell of mass m and radius r starts from rest and rolls without slipping down the track shown in Fig. 10.57. Points A and B are on a circular part of the track having...
-
Uhura Resort opened for business on June 1 with eight air-conditioned units. Its trial balance on August 31 is as follows. Other data:1. The balance in prepaid insurance is a one-year premium paid on...
-
Good Times Express Company had retained earnings of \(\$ 160\) million at December 31, 2006. The company reported these figures for 2007: Requirement Beginning with net income, prepare a combined...
-
Cordelia Barrera is attempting to monitor a filling process that has an overall average of 705 cc. The average range is 6 cc. If you use a sample size of 10, what are the upper and lower control...
-
Discuss the interdisciplinary nature of civil engineering research and practice, highlighting the integration of concepts from fields such as materials science, computer science, and urban planning...
-
Determine expressions for the mean residence time given "outward" diffusional release into a perfect sink from a) A cylindrical monolithic device of radius a, with ends capped. b) A spherical device...
-
1. For the company IKEA describe the importance/derivation of the company's size, revenue, market-share, reputation, dates, ethics and stakeholders. Try to be as specific as possible and give...
-
A 22.7-g sample of diatomic oxygen gas molecules is in a sealed rigid container that has a volume of \(0.0240 \mathrm{~m}^{3}\). If the gas is initially at \(-1.50^{\circ} \mathrm{C}\), by how much...
-
An ideal gas undergoes an isentropic process that changes its pressure from \(P_{i}\) to \(P_{f}\) and its volume from \(V_{\mathrm{i}}\) to \(V_{t}\). What is the work done on the gas?
-
An iceberg initially at \(273 \mathrm{~K}\) is stopped in its motion by a small island. During the first few seconds of contact, \(10 \mathrm{~kJ}\) of energy is transferred thermally from the island...
-
At room temperature, do collisions between nitrogen molecules have enough energy to set a nitrogen molecule rotating? Enough to set it vibrating? The quantum of energy associated with rotation is...
-
Derive an expression for converting a temperature (a) from the Celsius scale to the Fahrenheit scale, (b) from the Kelvin scale to the Fahrenheit scale, and (c) from the Fahrenheit scale to the...
-
The given two points are (-5,0) and (1,3) Then we have to find the distance between them.
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
The Assembly Department produced 2,000 units of product during June. Each unit required 1.75 standard direct labor hours. There were 3,800 actual hours used in the Assembly Department during June at...
-
Elegant Occasions Inc. sells china and flatware over the Internet. For the next period, the budgeted cost of the sales order processing activity is $78,300, and 4,350 sales orders are estimated to be...
-
Barley Medical Center has a single operating room that is used by local physicians to perform surgical procedures. The cost of using the operating room is accumulated by each patient procedure and...
-
A steel cylinder containing air has a closely fitted piston and a set of stops as shown in Fig. 3.7. The piston is loaded with certain weights. The air inside the cylinder is initially at...
-
What is the difference between compressed liquid and saturated liquid?
-
Explain the significance of critical point and triple point.

Study smarter with the SolutionInn App


