Question: HC CH HC CH The given molecules are (a) constitutional isomers (b) conformers (c) stereoisomers (d) identical (e) None of these
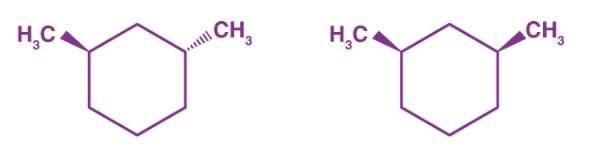

HC CH HC CH The given molecules are (a) constitutional isomers (b) conformers (c) stereoisomers (d) identical (e) None of these
Step by Step Solution
★★★★★
3.31 Rating (160 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

The detailed ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


