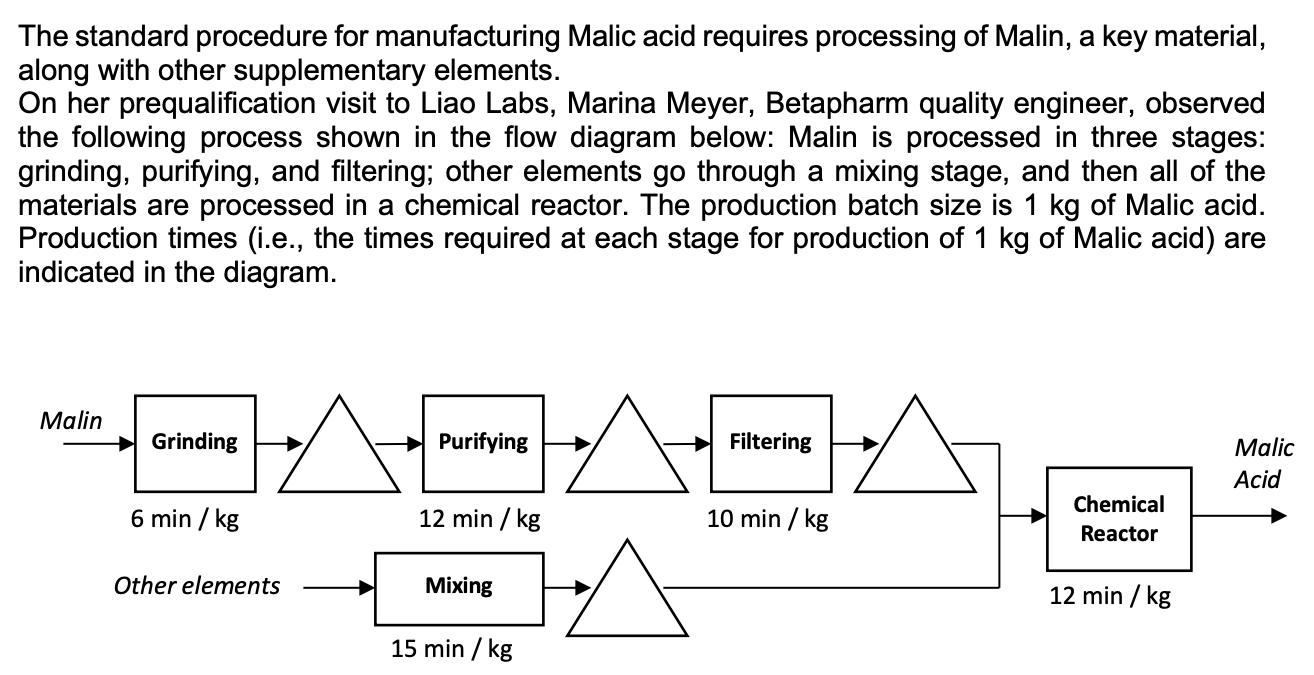
The standard procedure for manufacturing Malic acid requires processing of Malin, a key material, along with...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
The standard procedure for manufacturing Malic acid requires processing of Malin, a key material, along with other supplementary elements. On her prequalification visit to Liao Labs, Marina Meyer, Betapharm quality engineer, observed the following process shown in the flow diagram below: Malin is processed in three stages: grinding, purifying, and filtering; other elements go through a mixing stage, and then all of the materials are processed in a chemical reactor. The production batch size is 1 kg of Malic acid. Production times (i.e., the times required at each stage for production of 1 kg of Malic acid) are indicated in the diagram. Malin Grinding Purifying Filtering Malic Acid Chemical 6 min / kg 12 min / kg 10 min / kg Reactor Other elements Mixing 12 min / kg 15 min / kg (a) Based on Marina's observations, determine Liao's capacity rate for producing Malic acid. (b) An important capability that Betapharm desires is the ability to fill rush orders. How long would it take for Liao to fill a rush order of 100 kg? (c) While at Liao, Marina furrowed her brow as Mr. Liao, the owner, showed her Liao Lab's capacity numbers. Mr. Liao mentioned that he is considering purchasing a new machine to reduce the processing time of Purifying from 12 minutes to 6 minutes per kg. If this change is made, how will your answer to (a) and (b) change? The standard procedure for manufacturing Malic acid requires processing of Malin, a key material, along with other supplementary elements. On her prequalification visit to Liao Labs, Marina Meyer, Betapharm quality engineer, observed the following process shown in the flow diagram below: Malin is processed in three stages: grinding, purifying, and filtering; other elements go through a mixing stage, and then all of the materials are processed in a chemical reactor. The production batch size is 1 kg of Malic acid. Production times (i.e., the times required at each stage for production of 1 kg of Malic acid) are indicated in the diagram. Malin Grinding Purifying Filtering Malic Acid Chemical 6 min / kg 12 min / kg 10 min / kg Reactor Other elements Mixing 12 min / kg 15 min / kg (a) Based on Marina's observations, determine Liao's capacity rate for producing Malic acid. (b) An important capability that Betapharm desires is the ability to fill rush orders. How long would it take for Liao to fill a rush order of 100 kg? (c) While at Liao, Marina furrowed her brow as Mr. Liao, the owner, showed her Liao Lab's capacity numbers. Mr. Liao mentioned that he is considering purchasing a new machine to reduce the processing time of Purifying from 12 minutes to 6 minutes per kg. If this change is made, how will your answer to (a) and (b) change?
Expert Answer:
Answer rating: 100% (QA)
Total time required to complete grinding purifying and filtering as 61210 28 min kg As the other pro... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
Malic acid is a weak diprotic organic acid with Ka1 = 4.0 104 and Ka2 = 9.0 106. a. Letting the symbol H2A represent malic acid, write the chemical equations that represent Ka1 and Ka2. Write the...
-
A chemical reactor process whose production rate is a function of catalyst addition is shown in block diagram form in Figure P10.7 [10]. The time delay is T = 50 s, and the time constant T is...
-
Liao Manufacturing produces two keyboards, one for laptop computers and the other for desktop computers. The production process is automated, and the company has found activity-based costing useful...
-
Hussein Hage has just approached a venture capitalist for financing for his new restaurant, Bistro Sally. The lender is willing to loan Bistro Sally Inc. $240,000 at a high-risk interest rate of 9%....
-
An insulated rigid tank is divided into two compartments of different volumes. Initially, each compartment contains the same ideal gas at identical pressure but at different temperatures and masses....
-
What are four [4] functions of powertrain control module (PCM) in electronic compression ignition engine management system?
-
Why does a use case need a unique, numeric identifier?
-
Evaluate the professional judgment used by Kang and the firm in assessing the client's accounting and reaching its own decision to accept it?
-
As discussed in Reasons to Be Cheerful "roughly a third of us are actively avoiding the news;", like ostriches sticking their heads in the sands, do you think this is a healthy approach to handling...
-
Cinder Inc. is a Canadian-controlled private corporation based in your province. The company operates a wholesale business. The following information is provided for its year ended May 31, 2020: 1....
-
Starbucks' decision to buy out its joint venture partner in Japan following several years of operation and instead establish a wholly owned subsidiary Multiple Choice suggests that the company no...
-
Next Ltd. has a $250,000, 15%, 5-year bond outstanding. Interest is paid semi-annually, and the required rate of return is 10%. What is the value of the bond if it's repayable at a premium of 10%?
-
Check the articles that talk about (possible shift) in Boeing's Operations Strategy. You are more than welcome to find more in the same alignment. Your main job is to try to compare and contrast. How...
-
Complete a written analysis of leadership within the film (use the topics you have covered in your readings, assignments. (100 POINTS) This assignment provides the opportunity to synthesize your...
-
Brexit and the UK Answer the following items in one paragraph or more and feel free to use additional resources in your research. Research the current GDP of the European Union and how it ranks in...
-
when considering recruiting by posting to social media, in the U.S. we need to be sensitive to possible discriminatory claims, would that also be true when recruiting in Europe?
-
Explain the cultural diversity issue that must be addressed when forming partnerships to prevent domestic violence.
-
A Bloomberg Businessweek subscriber study asked, In the past 12 months, when traveling for business, what type of airline ticket did you purchase most often? A second question asked if the type of...
-
Explain how x rays are produced when an electron beam falls on a metal target.
-
An experiment calls for 35.0 g of hydrochloric acid that is 20.2% HCl by mass. How many grams of HCl is this? How many grams of water?
-
Part 1: In an insulated container, you mix 200. g of water at 80oC with 100. g of water at 20oC. After mixing, the temperature of the water is 60oC. a. How much did the temperature of the hot water...
-
Consider expansion of a function in terms of a series \(F_{n}(x)\) in the following form: \[f(x)=\sum_{n} A_{n} F_{n}(x)\] If the functions \(F_{n}\) are orthogonal then this property helps to unfold...
-
A concrete wall \(20 \mathrm{~cm}\) thick is initially at a temperature of \(20^{\circ} \mathrm{C}\), and is exposed to steam at pressure \(1 \mathrm{~atm}\) on both sides. Find the time for the...
-
Analyze the transient problem with the Dirichlet condition for a long cylinder and for a sphere. Derive expressions for the eigenfunctions, eigenconditions, and eigenvalues. Find the series...

Study smarter with the SolutionInn App


