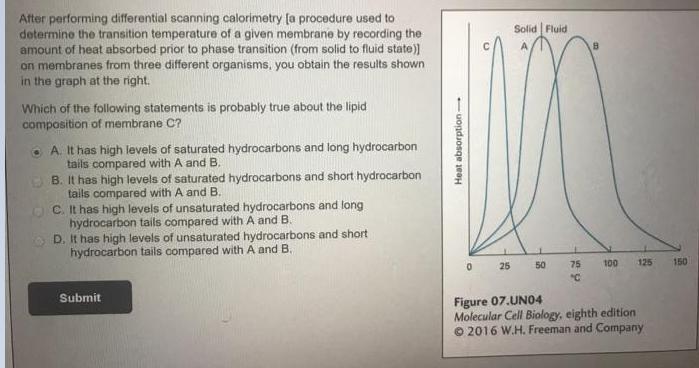
After performing differential scanning calorimetry (a procedure used to determine the transition temperature of a given...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
After performing differential scanning calorimetry (a procedure used to determine the transition temperature of a given membrane by recording the amount of heat absorbed prior to phase transition (from solid to fluid state) on membranes from three different organisms, you obtain the results shown in the graph at the right. Solid Fluid Which of the following statements is probably true about the lipid composition of membrane C? A. It has high levels of saturated hydrocarbons and long hydrocarbon tails compared with A and B. B. It has high levels of saturated hydrocarbons and short hydrocarbon tails compared with A and B. C. It has high levels of unsaturated hydrocarbons and long hydrocarbon tails compared with A and B. O D. It has high levels of unsaturated hydrocarbons and short hydrocarbon tails compared with A and B. 25 50 75 100 125 150 "C Submit Figure 07.UN04 Molecular Cell Biology, eighth edition © 2016 W.H. Freeman and Company Heat absorption After performing differential scanning calorimetry (a procedure used to determine the transition temperature of a given membrane by recording the amount of heat absorbed prior to phase transition (from solid to fluid state) on membranes from three different organisms, you obtain the results shown in the graph at the right. Solid Fluid Which of the following statements is probably true about the lipid composition of membrane C? A. It has high levels of saturated hydrocarbons and long hydrocarbon tails compared with A and B. B. It has high levels of saturated hydrocarbons and short hydrocarbon tails compared with A and B. C. It has high levels of unsaturated hydrocarbons and long hydrocarbon tails compared with A and B. O D. It has high levels of unsaturated hydrocarbons and short hydrocarbon tails compared with A and B. 25 50 75 100 125 150 "C Submit Figure 07.UN04 Molecular Cell Biology, eighth edition © 2016 W.H. Freeman and Company Heat absorption
Expert Answer:
Answer rating: 100% (QA)
Answer Correct Option C It has high levels of unsaturated hydrocarbons and long hydrocarbon tails co... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The results of a trial used to determine the capabilities of a new diagnostic test are shown in Table 9. Use the empirical probabilities obtained from the table to calculate the probability that a...
-
A Knudsen cell was used to determine the vapour pressure of germanium at 1000C. During an interval of7200 s the mass loss through a hole of radius 0.50 mm amounted to 43 ug, what is the vapour...
-
A magnetic field can be used to determine the sign of charge carriers in a current- carrying wire. Consider a wide conducting strip in a magnetic field oriented as shown in Fig. 19.34. The charge...
-
Calculate the standard entropy change for the following reactions at 25C. Comment on the sign of r S. (a) 2 Al(s) + 3 Cl 2 (g) 2 AlCl 3 (s) (b) 2 CH 3 OH() + 3 O 2 (g) 2 CO 2 (g) + 4 H 2 O(g)
-
Choose a business that you would like to own and write a short description of that business, including the business name, the products or services it would sell, and the types of customers the...
-
Explain the decision process used to encourage high-quality decision-making. Discuss how the concepts of audit evidence, uncertainty, risk, and professional skepticism affect the approach to...
-
Two batteries supply current to the circuit in Figure P23.20. The figure shows the potential difference across two of the resistors and the value of the third resistor. What current is supplied by...
-
The energy transferred from the anterior chamber of the eye through the cornea varies considerably depending on whether a contact lens is worn. Treat the eye as a spherical system and assume the...
-
An investment of $ 40,000 is estimated to yield $ 43,000 at the end of the first year, $ 79,000 in the middle of the second year and $ 24,000 at the end of the second year. The return requirement for...
-
Summers and Dooley are partners in a small business, "Summers & Dooley Co." The two partners disagree about whether or not to expand the business; Dooley wants to expand and is willing to take the...
-
A Canadian company borrowed US$1,000,000 on July 1, 2017, with interest at 6% to be paid on December 31 and June 30 each year of the five year term of the loan. The company prepares financial...
-
Explain how the managers in the story could be better prepared for these cultural differences. 3. Present three differences in business culture the US manager is likely to encounter in your peer's...
-
Crane Orthotics Company distributes a specialized ankle support that sells for $50. The company's variable costs are $35 per unit; fixed costs total $380,000 each year. (a1) Your answer is incorrect....
-
The division manager of Division B received the following operating income data for the past? year: Division B of Deisel, Inc. Income Statement For the Year Ended December 31, 2018 Product Line T205...
-
1. Calculate the following risk ratios for 2021 and 2022: (Round your answers to 1 decimal place.) 2. Calculate the following profitability ratios for 2021 and 2022: (Round your answers to 1 decimal...
-
The A & Co received a crossed check amounting to $5,000 from the B & Co. The A & Co. deposited the check into bank on the same date on which it was received. The amount of the check from B & Co would...
-
What line of code will create the following: mySwitches LightSwitch[] Ref null O LightSwitch [5] mySwitches = new LightSwitch[]; O LightSwitch] mySwitches = new LightSwitch[5]: O LightSwitch[5]...
-
Write a paper about the Working relationship in the organization- collaboration within and outside the organization
-
The energy levels of a CH3 group attached to a larger fragment are given by the expression for a particle on a ring, provided the group is rotating freely. What is the high-temperature contribution...
-
The Li2+ion is hydrogenic and has a Lyman series at 740 747 cm3, 877 924 cm-1, 925 933 cm-1, and beyond. Show that the energy levels are of the form -hcRln2 and find the value of R for this ion. Go...
-
A piece of zinc of mass 5.0 g is dropped into a beaker of dilute hydrochloric acid. Calculate the work done by the system as a result of the reaction. The atmospheric pressure is 1.1 atm and the...
-
Draw a block diagram for the feedback control of a liquid-level system, which consists of a valve with a control knob (0\%-100\%) and a liquid-level sensor. Clearly label essential components and...
-
A control system is represented using the block diagram shown in Figure 10.59. Sketch the root locus with respect to the proportional control gain K. Determine all the values of \(K\) for which the...
-
Roughly sketch the root locus with respect to \(K\) for the equation of \(1+K L(s)=0\) and the following choices for \(L(s)\). Make sure to give the asymptotes, arrival or departure angles, and...

Study smarter with the SolutionInn App


