Question: help please Draw the structure of the compound identified by the following simulated H and 13C NMR spectra. The molecular formula of the compound is
help please
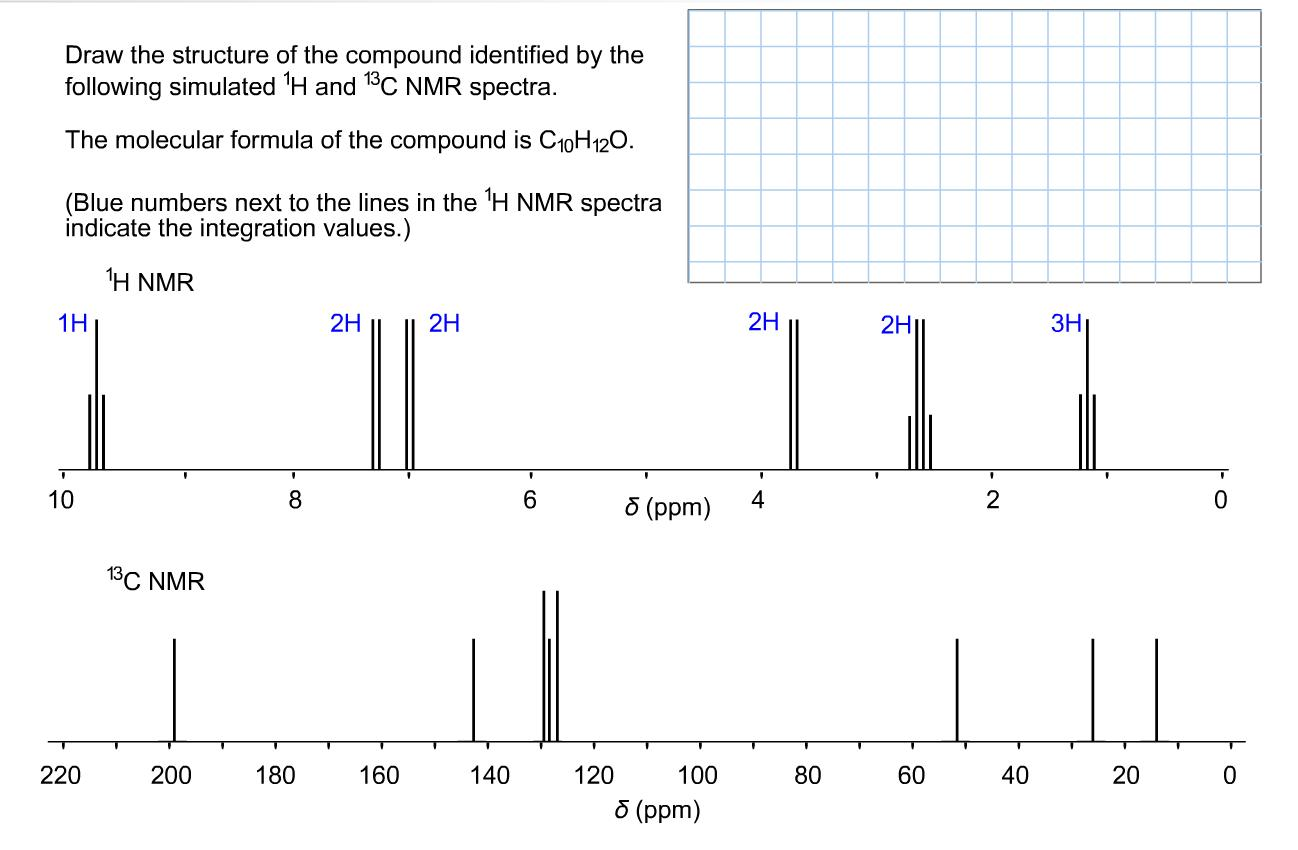
Draw the structure of the compound identified by the following simulated H and 13C NMR spectra. The molecular formula of the compound is C10H12O. (Blue numbers next to the lines in the H NMR spectra indicate the integration values.) H NMR 1H 1 10 220 13C NMR 200 8 180 2H 160 2H 140 6 120 8 (ppm) 100 5 (ppm) 2H 4 80 2H1 60 2 40 3H T 20 0 0
Step by Step Solution
★★★★★
3.46 Rating (166 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Based on the provided information the compound has a molecular formula of C120H ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


