Aspirin, an acetyl derivative of salicylic acid, is a white, crystalline, weakly acidic substance, with a...
Fantastic news! We've Found the answer you've been seeking!
Question:

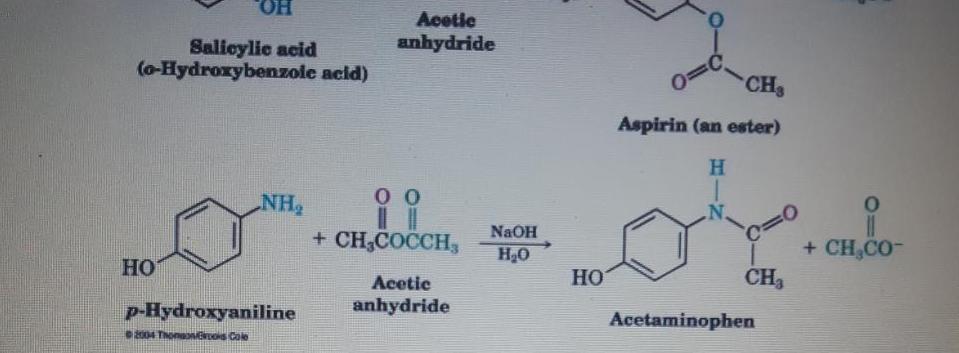
Transcribed Image Text:
Aspirin, an acetyl derivative of salicylic acid, is a white, crystalline, weakly acidic substance, with a melting point of 136 "C (277 F), and a boiling point of 140 °C (284 °F).24 Its acid dissociation constant (pK.) is 3.5 at 25 °C (77 °F) (0) With reference to electron withdrawing and releasing group effects explain why Aspirin is stronger acid than Acetaminophen. (3marks) Acetaminophen undergoes reduction reaction to give product A. Illustrate the (I) complete reaction including the reagent and condition involved. (3marks) Aspirin reacts with Grignard reagent CH3M9B to give Structure 'B'. Show the (ii) formation of structure B. (3marks) Using chemical equation give the formation of product when p-Hudroxwaniline (2 marks) (iv) reacts with bromine. HO. Acetic anhydride Salicylic acid (o-Hydroxybenzole acid) CH Aspirin (an ester) H. NH + CH,COCCH, %3D + CH,CO- NAOH H20 Acetic HO CH3 p-Hydroxyaniline anhydride Acetaminophen 2004 ThonaNos Cole Aspirin, an acetyl derivative of salicylic acid, is a white, crystalline, weakly acidic substance, with a melting point of 136 "C (277 F), and a boiling point of 140 °C (284 °F).24 Its acid dissociation constant (pK.) is 3.5 at 25 °C (77 °F) (0) With reference to electron withdrawing and releasing group effects explain why Aspirin is stronger acid than Acetaminophen. (3marks) Acetaminophen undergoes reduction reaction to give product A. Illustrate the (I) complete reaction including the reagent and condition involved. (3marks) Aspirin reacts with Grignard reagent CH3M9B to give Structure 'B'. Show the (ii) formation of structure B. (3marks) Using chemical equation give the formation of product when p-Hudroxwaniline (2 marks) (iv) reacts with bromine. HO. Acetic anhydride Salicylic acid (o-Hydroxybenzole acid) CH Aspirin (an ester) H. NH + CH,COCCH, %3D + CH,CO- NAOH H20 Acetic HO CH3 p-Hydroxyaniline anhydride Acetaminophen 2004 ThonaNos Cole
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Aspirin or acetyl salicylic acid is synthesized by reacting salicylic acid with acetic anhydride: (a) How much salicylic acid is required to produce 0.400 g of aspirin (about the content in a...
-
Aspirin (C9H8O4) is produced from salicylic acid (C7H6O3) and acetic anhydride (C4H6O3): C7H6O3 + C4H6O3 C9H8O4 + HC2H3O2 (a) How much salicylic acid is required to produce 1.5 102 kg of aspirin,...
-
Acetic anhydride is formed from acetic acid in a condensation reaction that involves the removal of a molecule of water from between two acetic acid molecules. Write the chemical equation for this...
-
According to Michel Foucault, while it is an advancement to no longer torture persons, modern forms of disciplinary punishment are sinister because they strive for _____________. a. routines. b....
-
A Philip Morris subsidiary, C. A. Tabacalera Nacional, and a B. A. T. subsidiary known as C. A. Cigarrera Bigott entered into a contract with La Fundacin del Nio (the Childrens Foundation) of...
-
Smith Company and Jones Company enter into an interest rate swap, with Smith paying fixed interest to Jones, and Jones paying floating interest to Smith. Smith now goes bankrupt and so defaults on...
-
These charged particles are traveling in circular orbits with velocities and field directions as noted. Which particles have a negative charge? x X x A. B. C. D.
-
Federal Agency Financial Statements. Using the data from Problem 112, prepare the following: a. In general journal form, entries to close the budgetary accounts as needed and to close the operating...
-
sheila pays corporate taxes. the girm has a debt equity ratio of 1. the pre tax cost of debt if 1.5% while the unlevered cost of capital is 10%. what is the approximate cost of equity if the tax rate...
-
Complete the statement of cash flow for January through June on the CashFlow worksheet by completing the following tasks. As part of the process, you will need to use a circular reference. You will...
-
Two thin rods each of mass m and length l are joined to form L shape as shown. The moment of inertia of rods about an axis passing through free end of a rod and perpendicular to the both the ends is
-
(a1, a2,..., Consider the following recursive algorithm, which takes as input a sequence , an) of n numbers, where n is a power of two, i.e., n = 2k for some integer k > 0: Algorithm MYSTERY (a1,...
-
A tower tower that is 200 m tall is leaning to one side. From a certain point on the ground on that side, the angle of elevation to the top of the tower is 70. From a point 55 m closer to the tower,...
-
In 1999, the population of Richmond-Petersburg, Virginia, metropolitan area, was 961,400 and was increasing at a at roughly 9200 people per year. The average annual income in the area was $30,593 per...
-
A taxpayer is making some changes to their home by installing some energy-efficient property in TY2023. Last year, they claimed $300 in nonbusiness energy property credit for similar changes. This...
-
Given two sorted lists A[1..n] and B[1..n]. We would like to find the median of the union of the two lists. For simplicity, assume that the union of A and B does not contain any duplicate items and...
-
Write the necessary commands to program the Nablus router using the protocol OSPF for this ip 2 0 . 0 . 0 . 0 / 8
-
Nike manufactures shoes and sportswear. How has the Internet changed the way this company communicates with its suppliers and retail customers?
-
Consider the format ion, HCO2-, which is the anion formed when formic acid loses an H+ ion. The H and the two O atoms are bonded to the central C atom. (a) Write one or more appropriate Lewis...
-
Write the balanced chemical equations for (a) The complete combustion of acetic acid (CH3COOH), the main active ingredient in vinegar (b) The decomposition of solid calcium hydroxide into solid...
-
The Henry's law constant for helium gas in water in 30 oC is 3.7 10-4 M/atm and the constant for N2 at 30 oC is 6.0 10-4 M/atm. If the two gases are each present at 1.5 atm pressure, calculate the...
-
Explain the difference between idiosyncratic and systematic risks.
-
Demonstrate how to reduce risk through hedging and diversification.
-
Assume that the economy can experience four possible states: high growth, normal growth, recession, or depression. For each of those states, you expect the following stock market returns for the...

Study smarter with the SolutionInn App



