How many atoms are present in each of the following samples? a) 0.386 mol of HO...
Fantastic news! We've Found the answer you've been seeking!
Question:

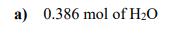
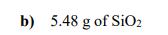
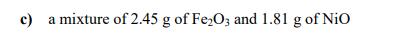
Transcribed Image Text:
How many atoms are present in each of the following samples? a) 0.386 mol of H₂O b) 5.48 g of SiO₂2 c) a mixture of 2.45 g of Fe₂O3 and 1.81 g of NiO How many atoms are present in each of the following samples? a) 0.386 mol of H₂O b) 5.48 g of SiO₂2 c) a mixture of 2.45 g of Fe₂O3 and 1.81 g of NiO
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many rings are present in each of the following compounds? Each consumes 2 moles of hydrogen on catalytic hydrogenation. (a) C8H8 (b) C8H8CI2 (c) C8H8O (d) C8H10O2 (e) C8H9ClO
-
How many asymmetric carbon atoms are present in each of the following compounds? (a) (b) (c) H-C-C-C-CI H CI H OH CH CH2OH OH H H OH
-
How many unpaired electrons are present in each of the following in the ground state: O, O+, O-, Os, Zr, S, F, Ar?
-
Write each number in scientific notation. 0.875
-
J&W Buffet Co. employees earned $350,000 in the week ended December 17, 2010. Of this, $26,775 was deducted from employees pay for FICA and $62,000 was deducted for income taxes. Prepare the journal...
-
Based on the MO discussion of NH 3 in the text, find the average NH bond order in NH 3 by calculating the net number of bonds and dividing by the number of NH groups.
-
Knoop Harness Number (KHN) is equal to: (a) \(\frac{P}{L C}\) (b) \(\frac{P}{L^{2} C}\) (c) \(\frac{2 P}{L C}\) (d) \(\frac{2 P}{L^{2} C}\)
-
Weighted-average method, equivalent units. Consider the following data for the assembly division of Stiles Watches, Inc. The assembly division uses the weighted-average method of process costing....
-
Question 3 i. In the evaluation of construction companies financial statements the gearing ratio and liquidity ratio have been described as providing useful information on company's activities. What...
-
Murphy Delivery Service completed the following transactions during December 2018: Dec. 1 Murphy Delivery Service began operations by receiving $13,000 cash and a truck with a fair value of $9,000...
-
Assume that 107.62 yen equal $1. Also assume that 7.5415Skr equal $1. How many Japanese yen can you acquire in exchange for 6,200 Swedish krona? A. 419 yen B. 434 yen C. 41,719 yen D. 46,757 yen E....
-
Scenario 1 Marcus is on placement in your school whilst studying for his level 2 qualification. When he arrived at the school this morning he noticed that the outside climbing frame was very wet and...
-
Please provide a journal entry for this transaction ES es are charged on ted goods. Children's books are subject to e HST at 13% is les of other ms. 18 19 17 It Again 4-A Charity Blvd. Toronto, ON...
-
Every year Rick contributes 3% of his annual $32,500 salary to a retirement fund. His company contributes an amount equal to 1% of his salary.
-
Assume that the equation for demand for bread at a small bakery is Q-60-10P+4Y, where Q is the quantity of bread demanded in loaves and Y is the average income in the town in thousands of dollars....
-
The following is the information: Plan: Mean Expense Percentage of Free Plan Free 42.2 - 25% 28.4 67% 50% 13.1 33% 95% 18.1 43% The table is provided above I am asked to calculate the arc elasticity...
-
Suppose you are an investor based in Japan. You have invested in the common stocks of publicly listed firms in several countries. You plan to liquidate your holdings of the equity investments in...
-
Floyd Distributors, Inc., provides a variety of auto parts to small local garages. Floyd purchases parts from manufacturers according to the EOQ model and then ships the parts from a regional...
-
Sodium hydroxide is hygroscopic-that is, it absorbs moisture when exposed to the atmosphere. A student placed a pellet of NaOH on a watch glass. A few days later, she noticed that the pellet was...
-
A phase diagram of water is shown at the end of this problem. Label the regions. Predict what would happen as a result of the following changes: (a) Starting at A, we raise the temperature at...
-
Assuming that air contains 78 percent N2, 21 percent O2, and 1 percent Ar, all by volume, how many molecules of each type of gas are present in 1.0 L of air at STP?
-
Solve the matrix equation \([\mathbf{K}]\{\mathbf{Q}\}=\{\mathbf{F}\}\) using forward reduction and backward substitution, where, 5-10 [K] = -13-1 0-1 1 {F}= 0
-
Volunteer work can improve your skills and enhance your rsum. With a partner, seek volunteer opportunities on your campus or in your city. Make a list of volunteer groups that may need help. Here are...
-
What are six brainstorming questions to consider before starting your proposal?

Study smarter with the SolutionInn App



