How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg...
Fantastic news! We've Found the answer you've been seeking!
Question:
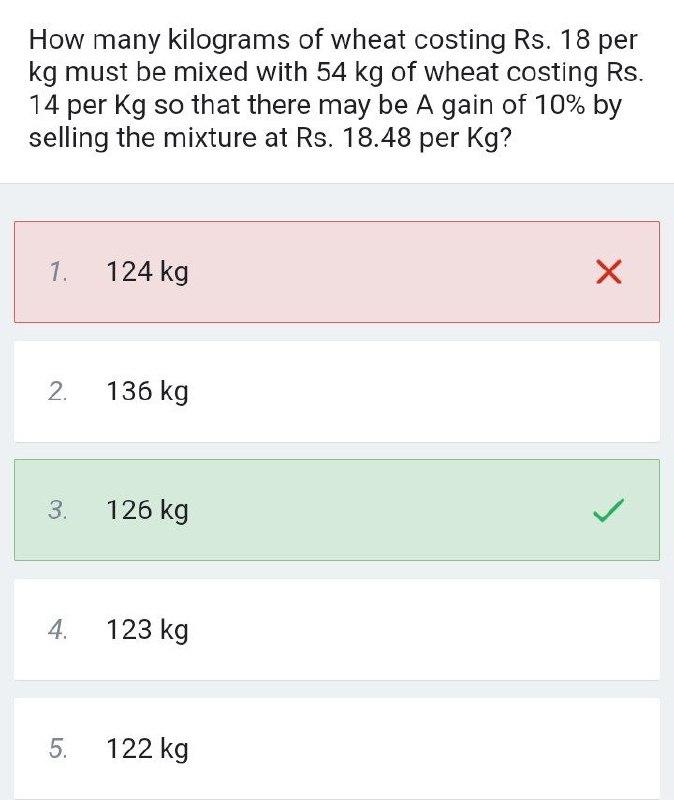
Transcribed Image Text:
How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg of wheat costing Rs. 14 per Kg so that there may be A gain of 10% by selling the mixture at Rs. 18.48 per Kg? 1. 124 kg 2. 136 kg 3. 126 kg 4. 123 kg 122 kg 5. How many kilograms of wheat costing Rs. 18 per kg must be mixed with 54 kg of wheat costing Rs. 14 per Kg so that there may be A gain of 10% by selling the mixture at Rs. 18.48 per Kg? 1. 124 kg 2. 136 kg 3. 126 kg 4. 123 kg 122 kg 5.
Expert Answer:
Answer rating: 100% (QA)
Solution 3 Type I wheat Rs 18kg Type II wheat Rs 14kg 3D Qu... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
How many kilograms of water must be processed to obtain 2.0 L of D2 at 25C and 0.90 atm pressure? Assume that deuterium abundance is 0.015 percent and that recovery is 80 percent.
-
How many kilograms of nickel must be added to 1.75 kg of copper to yield a liquids temperature of 1300(C?
-
How many kilograms of nickel must be added to 5.43 kg of copper to yield a solidus temperature of 1200(C?
-
On a cold winter day, the atmospheric temperature is - (on Celsius scale) which is below 0C. A cylindrical drum of height h made of a bad conductor is completely filled with water at 0C and is kept...
-
Define impasse, mediation, and strike, and explain the techniques that are used to overcome an impasse.
-
2. Interrupt and polling are useful when interfacing I/O devices that provide or require data at relatively low data transfer rates. a) Differentiate between the interrupt and polling. (4 marks) b)...
-
Find the values of \(k_{2}\) and \(m_{2}\) in Problem 9.54 in order to have the natural frequencies of the system at least 30 percent away from the forcing frequency. Data From Problem 9.54:- An...
-
Craig Company asks you to review its December 31, 2014, inventory values and prepare the necessary adjustments to the books. The following information is given to you. 1. Craig uses the periodic...
-
Southeast U\'s campus book store sells course packs for $15.00each, the variable cost per pack is $12.00, fixed costs for thisoperation are $300,000, and annual sales are 95,000 packs. The...
-
In a paint mixing plant, two tanks supply fluids to a mixing cistern. The height, h, of the fluid in the cistern is dependent upon the difference between the input mass flow rate, q, and the output...
-
An executive summary should be a brief outline of a proposal or longer document. Why? The summary is written for executives, and people in charge are busy. They demand a report that's concise....
-
When might a patients favorite color or high school alma mater be considered health information?
-
You believe that cooperation between the clinic receptionists and HIM staff would improve if phone responsibilities were more clearly defined. To whom would you assign the task of defining roles and...
-
The HIM staff tally information about the causes of complaints. What improvement tool would you use to prioritize the problems? Imagine you are the supervisor of the health information management...
-
After redesigning the process for investigating insurance denials, you want to monitor the effectiveness of your actions. What improvement tool would you use to determine whether the number of...
-
On 1 January 2010, an entity issued EUR 400,000 of 7 per cent bond at par. Interest on this loan stock is payable on 31 December each year. The stock is due for redemption at par on 31 December 2013...
-
Table 2.4 in Berk and DeMarzo (2020) provide key financial ratios for large US firms updated in Spring of 2018. Within this table, the authors highlight the 25%, median, and 75% quartiles for each...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Which of the following reagents can oxidize H2O to O2(g) under standard-state conditions? H+(aq),Cl-(aq), Cl2(g), Cu2+(aq), Pb2+(aq), MnO4- (aq) (in acid).
-
Which of the following metals can react with water? (a) Au, (b) Li, (c) Hg, (d) Ca, (e) Pt.
-
Consider the reaction Comment on the changes in the concentrations of SO2, O2, and SO3 at equilibrium if we were to (a) Increase the temperature; (b) Increase the pressure; (c) Increase SO2; (d) Add...
-
Form small groups of four to five individuals. Using the worksheet Be the Best We Can Be Team Norms, discuss how the answers to the questions can assist the group with developing team norms so that...
-
Explain why teams and groups are not the same.
-
List the teams of which you are a member. Select one of these teams to analyze. Is it a high performing team? If so, why is it? If not, why isnt it? What changes need to be made to increase the the...

Study smarter with the SolutionInn App


