64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A....
Fantastic news! We've Found the answer you've been seeking!
Question:
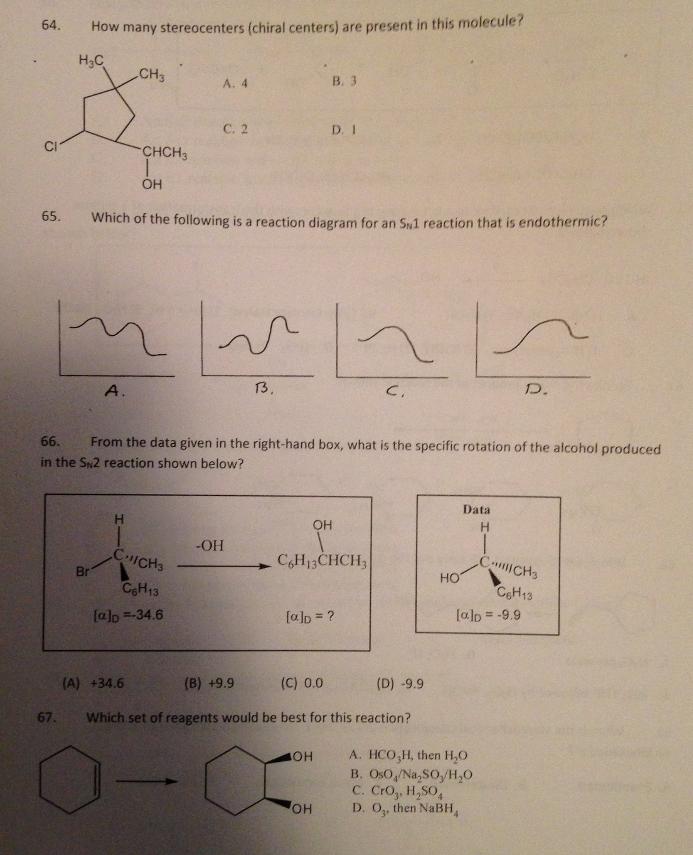
Transcribed Image Text:
64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO, 64. How many stereocenters (chiral centers) are present in this molecule? H3C CH3 B. 3 A. 4 С. 2 D. I CI CHCH3 65. Which of the following is a reaction diagram for an S1 reaction that is endothermic? A. 13. D. 66. From the data given in the right-hand box, what is the specific rotation of the alcohol produced in the Sn2 reaction shown below? Data OH -OH CHICHS C,H13CHCH, CICH3 HO Br CH13 CeH13 [alp =-34.6 [alp = ? lalo = -9.9 %3D (A) +34.6 (B) +9.9 (C) 0.0 (D) -9.9 67. Which set of reagents would be best for this reaction? A. HCO,H, then H,0 B. OsO,/Na, SO,/H,0 C. Cro, H,SO, D. O, then NABH, OH HO,
Expert Answer:
Posted Date:
Students also viewed these chemistry questions
-
Which of the following is equivalent to u=-Cost+V3 sint ? a) u = 2 cos t + 3 1 b) u = - cost+ 3 c) u = 2 cos t 3 d) u = : 1 os t- 3 tho
-
Which of the following is not related to ethical requirements of auditors? a. Due care. b. Independence in appearance. c. Independence in fact. d. Professional judgment.
-
Which of the following is not required by generally accepted auditing standards? a. Written representations. b. Attorney letter. c. Management letter. d. Engagement letter.
-
Which of the following pairs of steps in the programming process is in the correct order? a. Code the program, plan the logic b. Test the program, translate it into machine language c. Put the...
-
Kim is an employee of Azure Corporation. In the current year, she receives a cash salary of $30,000 and also is given 10 shares of Azure stock for services she renders to the corporation. The shares...
-
You have been asked to investigate some cost problems in the Assembly Department of JeVo Electronics Inc., a consumer electronics company. To begin your investigation, you have obtained the following...
-
You run at \(4 \mathrm{~m} / \mathrm{s}\) in the same direction as a river flowing at \(2 \mathrm{~m} / \mathrm{s}\). This speed is enough to keep you chatting with your friend who is steering a slow...
-
Tiana Shar, the controller for Caesar Glassware Company, is in the process of analyzing the overhead costs for the month of November. She has gathered the following data for the month. Labor...
-
Exercise 1 Motion on a Ramp In this exercise you will roll a marble down a ramp and calculate its linear acceleration and velocity. You will then create graphs to illustrate the motion of the marble.
-
Cardinal Company is considering a five-year project that would require a $2,975,000 investment in equipment with a useful life of five years and no salvage value . The company's discount rate is 14%....
-
Suppose that tax accountants increase the price for their service by 20 percent. The short-run demand for their service is less elastic than the long-run demand because in the long run taxpayers will...
-
Write a memo to your CFO. Include discussion of each of these points: How the sale portion of the sale-leaseback transaction should be accounted for at the lease's inception. How the gain on the sale...
-
Durham, Inc. issued $500,000 of its 10-year zero-coupon bonds on January 1, Year 6, to yield 9%. The effective interest method is used. PV of $1 (9%) PV of an Annuity of $1 (9%) FV of $1 (9%) FV of...
-
Explain the below general Information Retrieval (IR) system architecture - user feedback Ranked documents The user User query Query operations Executable query Retrieval system Document collection...
-
A right-hand circularly polarized, uniform plane wave E,His obliquely incident on the planar interface between two non-magnetic dielectric media as shown in the above figure. It is observed that the...
-
Crest Company's production budget for May, June, and July calls for 900, 1,000, and 1,100 units, respectively. Budgeted unit product cost formulas include direct materials, $24 per unit; direct...
-
The trial balance of David Advertising at December 31, 2014 before adjustments and the data needed for the month-end adjustments follow (the accounting period is one month): Cash Accounts Receivable...
-
Assume Eq. 6-14 gives the drag force on a pilot plus ejection seat just after they are ejected from a plane traveling horizontally at 1300 km/h. Assume also that the mass of the seat is equal to the...
-
Using the data from Table 11.3, what is the covariance between the stocks of Alaska Air and Southwest Airlines? Alaska Southwest Ford General Microsoft HP Air Airlines Motor Kellogg Mills Volatility...
-
If investors are holding optimal portfolios, how will the portfolios of a more risk averse and a more risk-tolerant investor differ?
-
According to the managerial entrenchment theory, managers choose capital structures so as to preserve their control of the firm. On the one hand, debt is costly for managers because they risk losing...

Study smarter with the SolutionInn App


