HSO. H0 Me CH Me 3. Provide a reasonable and detailed mechanism for each of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
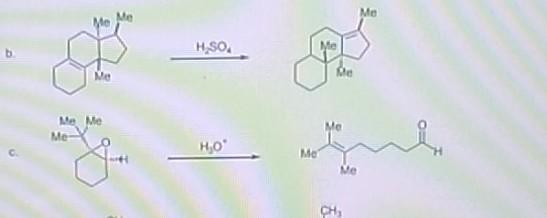

Transcribed Image Text:
H₂SO. H₂0° Me CH₂ Me 3. Provide a reasonable and detailed mechanism for each of the following carbocation rearrangements H₂SO. H₂0° Me CH₂ Me 3. Provide a reasonable and detailed mechanism for each of the following carbocation rearrangements
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Provide a detailed mechanism for each of the following reactions. (a) (b) (c) H,so, (cat.) OCH3 + CH OH (excess) 0 EtNH2 (excess) Cl NH H,O, H,SO, (cat.) OH
-
Draw a stepwise detailed mechanism for each of The following nucleophilic substitution reaction. Used curved arrows to show the movement of electrons, and draw the product including stereochemistry...
-
Provide a detailed mechanism for each of the following reactions. Include contributing resonance structures and the resonance hybrid for the arenium ion intermediates. (a) (b) (c) HNO HSO NO2 Br Bra,...
-
Metro builds and operates traditional shopping malls. It holds a $25 million deferred tax asset relating to credit carryforwards at the state, local, and Federal levels. No valuation allowances...
-
On April 16, 2014, ColorCo purchased a put option for $800 on Choco common stock. The put option gives ColorCo the option to sell 5,000 shares of Choco at a strike price of $25 per share for a period...
-
Data for Leeland Company are presented in P5-3B. In P5-3 Oct. 2 Purchased merchandise on account from Gregory Company at a cost of $35,000, terms 2/10, n/30, FOB destination. 4 The correct company...
-
What is an affirmative defense? Why is it important for the defendant to include affirmative defenses in the answer?
-
On March 1, a securities analyst recommended General Cinema stock as a good purchase in the early summer. The portfolio manager plans to buy 20,000 shares of the stock on June 1 but is concerned that...
-
two clerks are on call at FastService LTD. Customers arrive at a mean rate of 1 per minute, and, on average, it takes 2 minutes to serve a customer. It has been observed that, on average, customers...
-
Cost Center D within the Welding Department of the Mizer Corporation collected the following data during 2004. Cost Center D Overhead Expenses: \($4,800\) Cost Center D Direct Labor Hours: 2,800 Cost...
-
Differentiate between Strategic and Human UX Design? Discuss pros and cons of each?
-
The Farmer's Dairy Product case from the text (page 640 to 649) is the basis for this analysis. It is assumed that the costs structure for May 2021 is unchanged for the next three months. It is...
-
Consider two companies (A and B) that have been in business for the same amount of time (5 years) and they perform exactly equally with equal assets acquired on the same dates at the same costs. For...
-
A plastic rod of length d = 1.5 m lies along the x -axis with its midpoint at the origin. The rod carries a uniform linear charge density = 4.5 nC/m. The point P is located on the postive y -axis at...
-
Sweet Baking Inc. sells on credit terms of net 30. Its accounts are, on average, four days past due. If annual credit sales are $9.75 million, what is the company's balance sheet amount in accounts...
-
You are asked to design a cylindrical steel rod 60.0 cm long, with a circular cross section, that will conduct 140.0 J/s from a furnace at 420.0 C to a container of boiling water under 1 atmosphere...
-
You want to measure the speed of a 22 calibre bullet with a mass of 11 g, with only a spring, a block of mass 5 kg, a stopwatch, a ruler, a scale, and a frictionless table. You remember that you did...
-
H.J. Heinzs annual dividends were as follows: 1990 ..............$0.540 1991.............. 0.620 1992 .............. 0.700 1993.............. 0.780 1994 .............. 0.860 1995 .............. 0.940...
-
Open the energy-minimized 3D Molecular Models on the book's website for trans-1-tert- utyl-3-methylcyclohexane and trans-1,3-di-tert-butylcyclohexane. What conformations of cyclohexane do the rings...
-
Primary halides of the type ROCH2X apparently undergo SN1-type reactions, whereas most primary halides do not. Can you propose a resonance explanation for the ability of halides of the type ROCH2X to...
-
Compound M has the molecular formula C9H12. The 1H NMR spectrum of M is given in Fig. 14.29 and the IR spectrum in Fig. 14.30. Propose a structure for M.
-
Restore the file Boston Catering. Make the following adjustments and then print an income statement and balance sheet for the period 7/1/10 through 9/30/10 setting the columns to Month so that you...
-
Explain the journal entry method of recording end-of-period adjustments.
-
Give an example of an accrued expense other than the example given in this chapter. Explain how your example of accrued expense would be adjusted using journal entries.
Supply Chain Cost Control Using Activity Based Management 1st Edition - ISBN: 0849382157 - Free Book

Study smarter with the SolutionInn App



