Hydrogen peroxide in aqueous solution will decompose to produce oxygen gas and water. Use the graph...
Fantastic news! We've Found the answer you've been seeking!
Question:
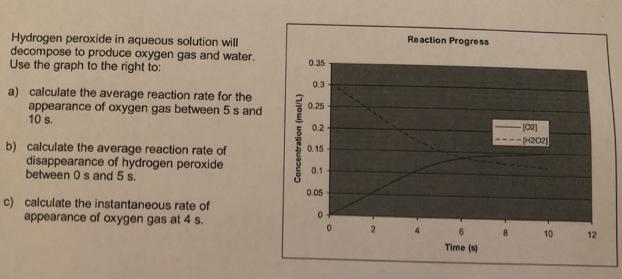
Transcribed Image Text:
Hydrogen peroxide in aqueous solution will decompose to produce oxygen gas and water. Use the graph to the right to: Reaction Progress 0.35 0.3 a) calculate the average reaction rate for the appearance of oxygen gas between 5 s and 10 s. 0.25 0.2 p42021 b) calculate the average reaction rate of disappearance of hydrogen peroxide between 0s and 5 s. 0.15 0.1 0.05 c) calculate the instantaneous rate of appearance of oxygen gas at 4 s. 10 12 Time (s) Concentration (mol/L) Hydrogen peroxide in aqueous solution will decompose to produce oxygen gas and water. Use the graph to the right to: Reaction Progress 0.35 0.3 a) calculate the average reaction rate for the appearance of oxygen gas between 5 s and 10 s. 0.25 0.2 p42021 b) calculate the average reaction rate of disappearance of hydrogen peroxide between 0s and 5 s. 0.15 0.1 0.05 c) calculate the instantaneous rate of appearance of oxygen gas at 4 s. 10 12 Time (s) Concentration (mol/L)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Hydrogen peroxide in aqueous solution decomposes by a first-order reaction to water and oxygen. The rate constant for this decomposition is 7.40 104/s. What quantity of heat energy is initially...
-
Hydrogen gas is evolved and an aqueous solution of aluminum chloride is formed when solid aluminum reacts with an aqueous solution of hydrochloric acid. Write the balanced chemical equation and...
-
Hydrogen gas combines spontaneously with oxygen gas to form water 2 H2 + O2 2 H2O Which term, enthalpy or entropy, predominates in the equation for the Gibbs free energy? How are the surroundings...
-
Suppose that we throw 30 fair dice. Recall that a fair die has six sides, numbered from 1 to 6, with each side occurring with equal probability. (a) (3pts) What is the probability that 5 of each of...
-
After the reunification of Germany in 1990, payments to rebuild the East led to a major expansion of aggregate demand in Gennany. The Gennan central bank responded by slowing money growth and raising...
-
Suppose that a Bayesian spam filter is trained on a set of 1000 spam messages and 400 messages that are not spam. The word "opportunity" appears in 175 spam messages and 20 messages that are not...
-
Stanford Seeles, engagement partner for the audit of the Torgesen-Tate Corporation, is planning the nature, timing, and extent of audit procedures for accounts payable. Prior year working papers...
-
A vapor mixture of n-butane (B) and n-hexane (H) contains 50.0 mole% butane at 120C and 1.0 atm, A stream of this mixture flowing at a rate of 150.0 L/s is cooled and compressed, causing some but not...
-
Payton took a friend for a birthday dinner. The total bill for dinner was $44.32 (including tax and a tip). If Payton paid a 19.6% tip, what was his bill before adding the tip? (Round your answer to...
-
It's amazing how much difference there is in the way proposals are presented at two different firms," said John Woods to his assistant, Pete Madsen, as he pointed to the stack of capital investment...
-
What force in lbf is exerted by the water on the bottom of a full rectangular storage tank that has a base of 1 0 ft x 1 7 ft and is 2 7 ft tall?
-
The decedent was married at the time of death. He was survived by his wife and children. The following were presented to you and you were asked to compute the 1) exclusive and conjugal properties...
-
(Figure: Demand for Coconuts) The accompanying figure shows the demand for coconuts. If coconuts are considered a normal good and there is an expectation on the part of consumers that the prices of...
-
The Solow Model in Reality Here is again the quick review of some algebraic properties of exponents: kakb=ka+b kak-b-ka-b k=1/kb kakbka-b k = k Ifk=a then k=a Questions 1-11 and A use the following...
-
10.2 What Trends Are Shaping Today's Labor Mark Explain the idea listed in each box: Larger, more diverse labor force Shift from manufacturing to service jobs Increase in knowledge workers...
-
Find the indefinite integral. Check your answer by differentiation. [2x(x +3) dx = +C
-
Problem 3 Assume an investor is holding a portfolio of six bonds investment characteristics of which are given in the table below: Bond Current Market Price Coupon, % Term to Maturity, years A 97.552...
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
Describe the primary structure of protein. What makes one protein different from another protein of the same size? What is the basis of the unique conformation of a protein?
-
The circumference of the earth at the equator is 40,000 km. This value is precise to two significant figures. Write this in scientific notation to express correctly the number of significant figures.
-
Magnesium nitride, Mg3N2, reacts with water to produce magnesium hydroxide and ammonia. How many grams of ammonia can you obtain from 7.50 g of magnesium nitride?
-
True or False. For an undamped system, the velocity leads the acceleration by \(\pi / 2\).
-
True or False. The motion diminishes to zero in both underdamped and overdamped cases.
-
True or False. The loss coefficient denotes the energy dissipated per radian per unit strain energy.

Study smarter with the SolutionInn App



