Hydrogen Peroxide is found in concentrations of 10-8 to 10-8 M in water droplets in the...
Fantastic news! We've Found the answer you've been seeking!
Question:
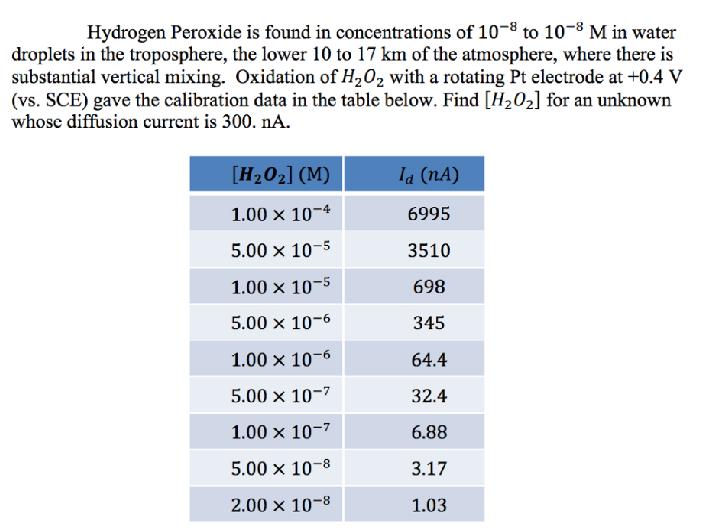
Transcribed Image Text:
Hydrogen Peroxide is found in concentrations of 10-8 to 10-8 M in water droplets in the troposphere, the lower 10 to 17 km of the atmosphere, where there is substantial vertical mixing. Oxidation of H,0, with a rotating Pt electrode at +0.4 V (vs. SCE) gave the calibration data in the table below. Find [H202] for an unknown whose diffusion current is 300. nA. [H202] (M) Ia (nA) 1.00 x 10-4 6995 5.00 x 10-5 3510 1.00 x 10-5 698 5.00 x 10-6 345 1.00 x 10-6 64.4 5.00 x 10-7 32.4 1.00 x 10-7 6.88 5.00 x 10-8 3.17 2.00 x 10-8 1.03 Hydrogen Peroxide is found in concentrations of 10-8 to 10-8 M in water droplets in the troposphere, the lower 10 to 17 km of the atmosphere, where there is substantial vertical mixing. Oxidation of H,0, with a rotating Pt electrode at +0.4 V (vs. SCE) gave the calibration data in the table below. Find [H202] for an unknown whose diffusion current is 300. nA. [H202] (M) Ia (nA) 1.00 x 10-4 6995 5.00 x 10-5 3510 1.00 x 10-5 698 5.00 x 10-6 345 1.00 x 10-6 64.4 5.00 x 10-7 32.4 1.00 x 10-7 6.88 5.00 x 10-8 3.17 2.00 x 10-8 1.03
Expert Answer:
Answer rating: 100% (QA)
The calibration curve is plotted between H 2 O 2 and I d The equation of best fit ... View the full answer

Posted Date:
Students also viewed these accounting questions
-
For the data given in table below find the best subset model using C, criterion and by using mean square residual criterion. Observation X1 X2 X3 X4 1 78 26 6. 60 2 74 1 29 15 22 104 11 56 8. 20 4 87...
-
A vertical rectangular water duct 1 m high and 0.1 m deep is placed in an environment of saturated steam at atmospheric pressure. If the outer surface of the duct is about 50?C, estimate the rate of...
-
A 4 m x 5 m x 6 m room contains 120 kg of air. Determine (a) Density. (b) Specific volume. (c) Mole (d) Specific molar volume of air. Assume molar mass of air to be 29 kg/kmol.
-
2. An analog baseband signal of bandwidth 125 kHz is quantized using a -law quantizer with -200. The signal is then sampled at a rate 3 times its Nyquist rate. The resulting data rate of the...
-
Consider the McDonalds tracking survey presented in Branding Brief 8-1. What might you do differently? What questions would you change or drop? What questions might you add? How might this tracking...
-
Describe the circumstances under which the current, quick, and cash ratios, respectively, are more appropriate measures of short-term liquidity than the other ratios?
-
(a) Draw an energy diagram for the situation shown in Figure 9.11 for the system that comprises Earth and the box. Assume the box keeps moving at constant velocity, and consider the rope to be part...
-
At the end of 2012, Sawyer Company is conducting an impairment test and needs to develop a fair value estimate for machinery used in its manufacturing operations. Given the nature of Sawyer??s...
-
The atomic number of element M, N, R, and T are 1, 8, 12 and 14 respectively. The element which can form ions by either losing or gaining electron(s) is M 11. A. B. N C R D. T 12. The atomic number...
-
Sofia Lofts case, but under a new set of assumptions on page 11 of the case. First re-read the Sofia Lofts case, then click here to download the new page 11. Specifically, you are to write up a brief...
-
In my Strategy class, we learned that the definition of strategy is'an approach to achieving a particular outcome.' The 'approach' involves answering two questions: (1) how will we compete and (2)...
-
The exhaust gas of a furnace burning a hydrocarbon fuel in air is sampled and found to be \(13.45 \% \mathrm{CO}_{2} ; 1.04 \% \mathrm{CO} ; 2.58 \% \mathrm{O}_{2} ; 7.25 \% \mathrm{H}_{2} \mathrm{O}...
-
A gas engine is operated on a stoichiometric mixture of methane \(\left(\mathrm{CH}_{4} ight)\) and air. At the end of the compression stroke the pressure and temperature are \(10 \mathrm{bar}\) and...
-
A mixture of propane and air with an equivalence ratio 0.9 (i.e. a weak mixture) is contained in a rigid vessel with a volume of \(0.5 \mathrm{~m}^{3}\) at a pressure of 1 bar and \(300...
-
A vessel contains a mixture of ethylene \(\left(\mathrm{C}_{2} \mathrm{H}_{4} ight)\) and twice as much air as that required for complete combustion. If the initial pressure and temperature are 5 bar...
-
In an experiment to determine the calorific value of octane \(\left(\mathrm{C}_{8} \mathrm{H}_{18} ight)\) with a bomb calorimeter the mass of octane was \(5.42195 \times 10^{-4} \mathrm{~kg}\), the...
-
Note: Please show the full calculation work clearly step by step to get the full marks. Calculate the values up to two decimal points. Write your response with a complete sentence in the space...
-
Planning: Creating an Audience Profile; Collaboration: Team Projects. Compare the Facebook pages of three companies in the same industry. Analyze the content on all available tabs. What can you...
-
Explain how the control variate technique is implemented.
-
Suppose that in September 2013 a company takes a long position in a contract on May 2014 crude oil futures. It closes out its position in March 2014. The futures price (per barrel) is \($88.30\) when...
-
A U.S. Treasury bond pays a 7% coupon on January 7 and July 7. How much interest accrues per \($100\) of principal to the bond holder between July 7, 2013, and August 9, 2013? How would your answer...

Study smarter with the SolutionInn App

