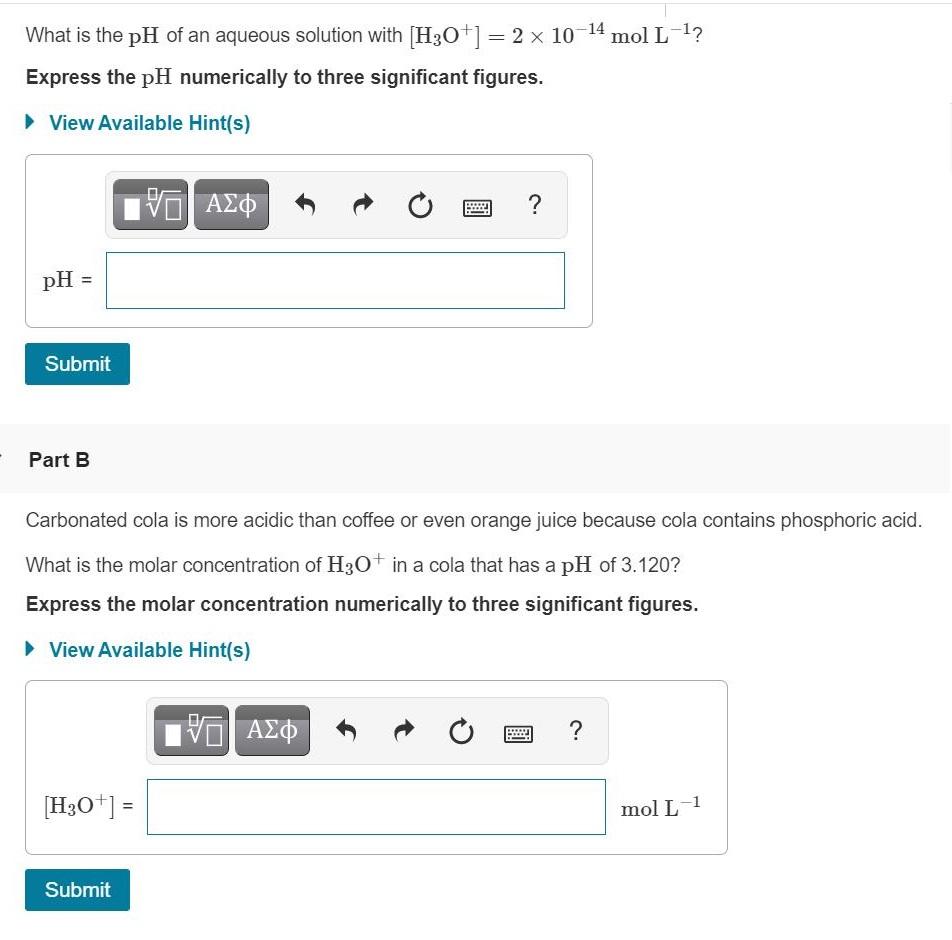
What is the pH of an aqueous solution with [H3O+|= 2 x 10 14 mol L-1?...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
What is the pH of an aqueous solution with [H3O+|= 2 x 10 14 mol L-1? Express the pH numerically to three significant figures. > View Available Hint(s) pH = Submit Part B Carbonated cola is more acidic than coffee or even orange juice because cola contains phosphoric acid. What is the molar concentration of H3O+ in a cola that has a pH of 3.120? Express the molar concentration numerically to three significant figures. > View Available Hint(s) [H3O*] = mol L-1 Submit What is the pH of an aqueous solution with [H3O+|= 2 x 10 14 mol L-1? Express the pH numerically to three significant figures. > View Available Hint(s) pH = Submit Part B Carbonated cola is more acidic than coffee or even orange juice because cola contains phosphoric acid. What is the molar concentration of H3O+ in a cola that has a pH of 3.120? Express the molar concentration numerically to three significant figures. > View Available Hint(s) [H3O*] = mol L-1 Submit
Expert Answer:
Answer rating: 100% (QA)
A Concentration of HO HO 2 10 molL since pH logHO so ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the pH of an aqueous solution of 3.6910 -3 M hydrobromic acid ? pH =
-
What is the pH of an aqueous solution of 4.8110 -3 M barium hydroxide ? pH =
-
What is the pH of an aqueous solution that is 0.0030 M pyruvic acid, HC3H3O3? (Pyruvic acid forms during the breakdown of glucose in a cell.)
-
CORPORATE FINANCE- RISK MANAGEMENT You manage a Swedish company that produces hats. You source your textiles mainly from Poland. The next input delivery from Poland is coming in three months. The...
-
Is the U. S. Court of Federal Claims a national court? Must a taxpayer go to Washington, D. C., to present a case to this court?
-
A triangular postage stamp is being designed such that the height h is 1.0 cm more than the base b. Find the possible height h such that the area of the stamp is at least 3.0 cm 2 .
-
Students who develop strong job-search skills have a career advantage. They develop clear career direction. They also communicate and promote their competitive edge to employers.
-
The following selected accounts appear in the ledger of Patton Environmental Inc. on July 1, 2012, the beginning of the current fiscal year: Preferred 2% Stock, $75 par (40,000 shares authorized,...
-
a. What is its value if the previous dividend was Do = $1.50 and investors expect dividends to grow at a constant annual rate of (1) -3%, (2) 0%, (3) 3%, or (4) 6%? Do not round intermediate...
-
SECTION B: ANSWER ANY THREE (3) QUESTIONS QUESTION TWO The following summarized statements of comprehensive income have been prepared for the year ended 31 December 2014 for Gold Reef Holdings and...
-
Workers compensations statutes a. provide benefits to employees injured in the course and scope of their work, unless the employee was at fault b. provide benefits to employees injured in the course...
-
how would I do what's in the highlighted area Your supervisor has asked you to complete a two-part project. The first part involves joining ServerSA1 to the company Active Directory domain, and the...
-
Company Z is looking at potentially introducing a new product, and has compiled the following annual estimates: Revenue: $ 3 0 , 0 0 0 Fixed Costs: $ 1 8 , 0 0 0 ...
-
1. Recall the "falling chain" problem from class and assume the chain to have length X and mass m. Y 9 In terms of the distance y, the chain's kinetic energy and potential energy are given by m 47 (A...
-
The following table shows the crude steel production in the USA, in millions of metric tons. Year Crude Steel Production 1980 0 80 1988 8 92 91 1991 11 (A) Find a quadratic function f(x) = ax2 + bx +...
-
Write C++ program that accepts coefficients of a quadratic equation from the user and displays the roots. The form of the quadratic equation is: y = ax +bx+c In this task we will assume that the...
-
Sigma Retail began operations on January 1, Year 1. Below are the transactions for the first year: Transaction # Date Transaction Description Amount 1 Jan 1, Y1 Issued common stock for cash $130,000...
-
Which of the following gives the range of y = 4 - 2 -x ? (A) (- , ) (B) (- , 4) (C) [- 4, ) (D) (- , 4] (E) All reals
-
Note whether hydrolysis occurs for each of the following ions. If hydrolysis does occur, write the chemical equation for it. Then write the equilibrium expression for the acid or base ionization...
-
Give the electron configurations of Cu+ and Cu2+.
-
Express the SI unit of energy as the product of two electrical units.
-
DNSE Inc. began operations in 2019. In its first year the company had a net operating loss of \(\$ 10,000\), which was carried forward and used to reduce income tax payable in 2020. In 2020, DNSE had...
-
The financial statements of Gibson Corporation for the first two years of operations reflected the following amounts. Assume a tax rate of \(25 \%\) for 2020 and 2021. Estimates of future earnings at...
-
Randolph Inc. considered the probability of a recent tax position taken related to the exclusion of certain revenues of \(\$ 10,000\) from taxable revenue. Randolph determined that this position is...

Study smarter with the SolutionInn App


