Question: This simple experiment on temperature measurement is aimed to compare and explain the measured temperature data from the standard temperatures of water. Under this

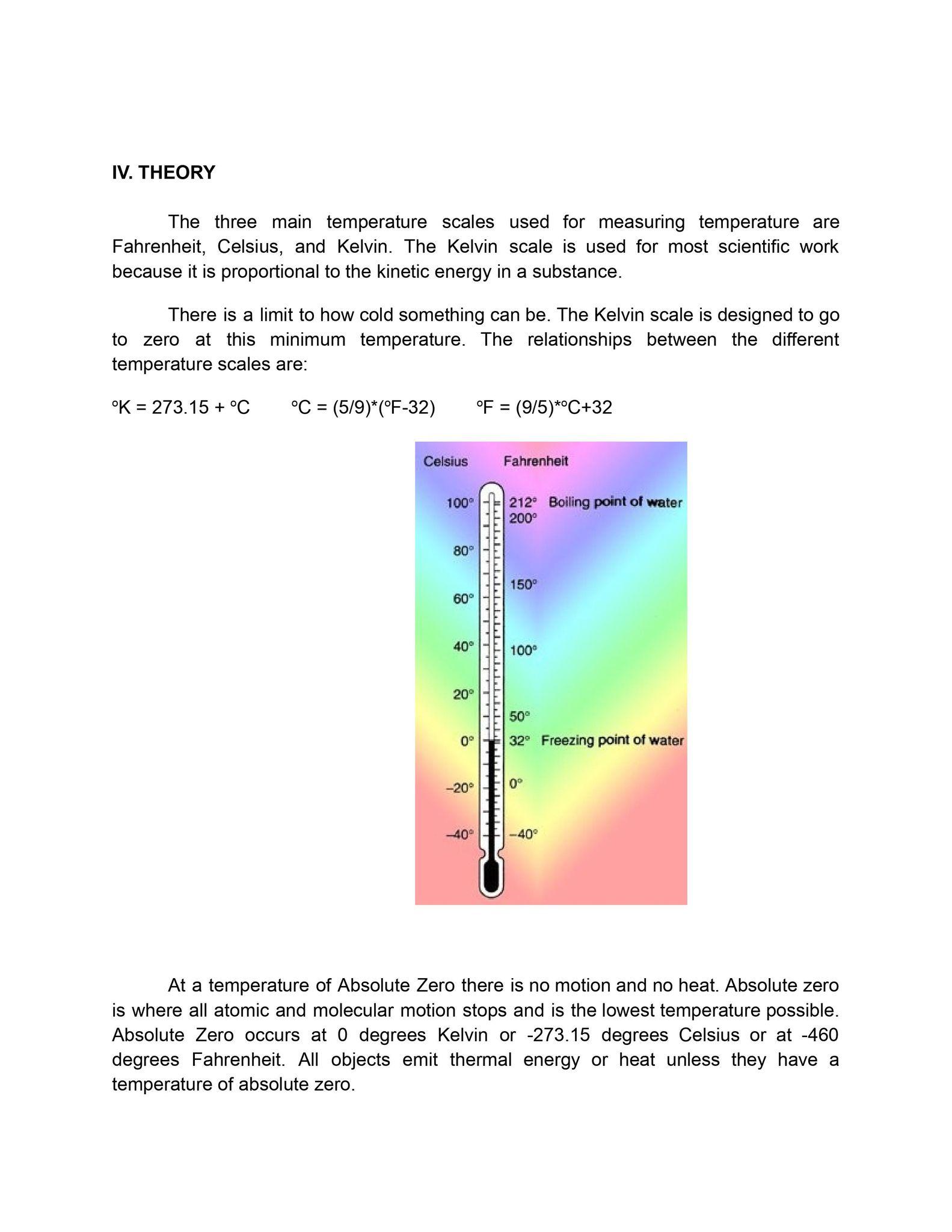


This simple experiment on temperature measurement is aimed to compare and explain the measured temperature data from the standard temperatures of water. Under this activity students will be able to write the experimental procedure. Explain the difficulty and errors in temperature measurement. Also, to introduce a new scale of temperature and correlate this to Fahrenheit and Celsius scales of temperature. III. INTRODUCTION Temperature is the most commonly measured parameter, yet in many respects it is the least understood. It is a surprisingly difficult parameter to measure with the precision that one might reasonably expect. To obtain accuracies better than 0.2C (0.4F) great care is needed. Errors occur due to the presence of temperature gradients, drafts, sensor nonlinearities, poor thermal contact, calibration drifts, radiant energy and sensor self heating. Generally the accuracy of all sensor types can be greatly improved by individual calibration. For more information, refer to the appropriate page on each sensor type. The information in this section is oriented towards electronic thermometers - those with an electrical output that can be connected to a measuring instrument, such as: a data acquisition system, a data logger, a control system or a chart recorder. However, there is also a wide range of thermometers that can be used for manual temperature measurement. These include: the glass thermometer, various gas thermometers, pressure based thermometers, bimetallic thermometers and temperature sensitive paint or film thermometers. IV. THEORY The three main temperature scales used for measuring temperature are Fahrenheit, Celsius, and Kelvin. The Kelvin scale is used for most scientific work because it is proportional to the kinetic energy in a substance. There is a limit to how cold something can be. The Kelvin scale is designed to go to zero at this minimum temperature. The relationships between the different temperature scales are: K = 273.15 + C = C (5/9)*(F-32) - F (9/5)*C+32 Celsius Fahrenheit 100 212 Boiling point of water 80 200 150 60 40 ------ 100 20 50 0 32 Freezing point of water -20 0 -40 -40 At a temperature of Absolute Zero there is no motion and no heat. Absolute zero is where all atomic and molecular motion stops and is the lowest temperature possible. Absolute Zero occurs at 0 degrees Kelvin or -273.15 degrees Celsius or at -460 degrees Fahrenheit. All objects emit thermal energy or heat unless they have a temperature of absolute zero. V. EXPERIMENTAL PROCEDURE 1. VII. TABULATION AND GRAPH VIII. DISCUSSION PART I. PRE-CALCULATION 1. Discuss the results of the conducted experiment. 2. Was the temperature of the boiling water bath exactly 100 deg C? Why or why not? 3. Was the temperature of the ice-water bath exactly 0 deg C? Why or why not? 4. Is temperature measurement difficult? 5. Discuss sources of error in temperature measurement. PART II. CALCULATION Introducing a new scale of temperature, say, degrees N. In this new scale of
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


