Identify the hybrid orbitals on all atoms in the moleculeCH 2 O. The H atoms in CH
Question:
Identify the hybrid orbitals on all atoms in the moleculeCH2O.
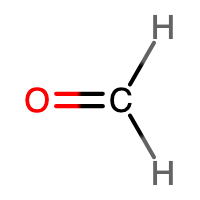
The H atoms in CH2O contain the following.
1 sp hybridorbital,1 sp2 hybrid orbital, 1 sp3 hybridorbital,2 sp hybrid orbitals,2 sp2 hybridorbitals,2 sp3 hybrid orbitals,3 sp hybrid orbitals,3 sp2 hybrid orbitals,3 sp3 hybrid orbitals, no hybridorbitals
The C atom in CH2O contains the following.
1 sp hybridorbital,1 sp2 hybrid orbital, 1 sp3 hybrid orbital,2 sp hybrid orbitals,2 sp2 hybrid orbitals,2 sp3 hybrid orbitals,3 sp hybrid orbitals,3 sp2 hybrid orbitals,3 sp3 hybrid orbitals, no hybridorbitals
The O atom in CH2O contains the following.
1 sp hybrid orbital,1 sp2 hybrid orbital, 1 sp3 hybrid orbital,2 sp hybrid orbitals,2 sp2 hybrid orbitals,2 sp3 hybrid orbitals,3 sp hybrid orbitals,3 sp2 hybrid orbitals,3 sp3 hybrid orbitals, no hybridorbitals

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward




