If a substance, at 1.0 atm of pressure is cooled from 400 K to 200K, (see...
Fantastic news! We've Found the answer you've been seeking!
Question:
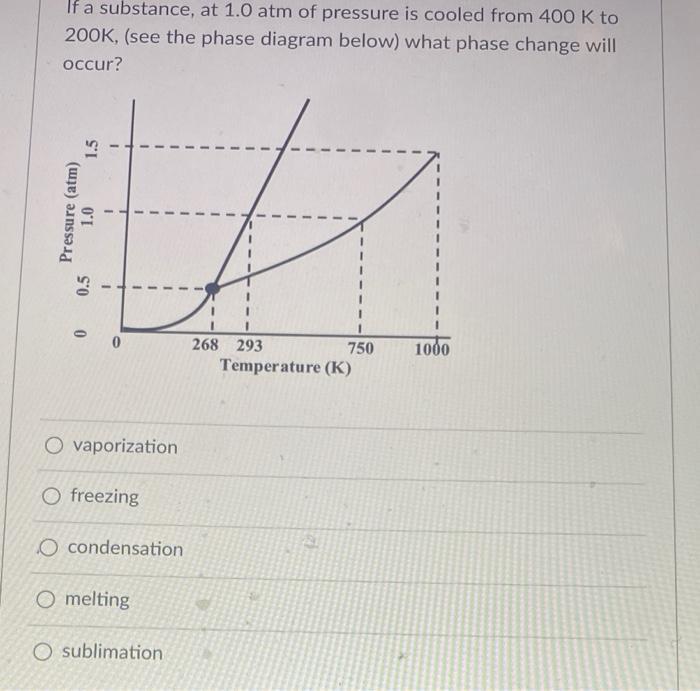
Transcribed Image Text:
If a substance, at 1.0 atm of pressure is cooled from 400 K to 200K, (see the phase diagram below) what phase change will occur? %3D 268 293 750 1000 Temperature (K) O vaporization O freezing O condensation O melting O sublimation Pressure (atm) 0.5 1.0 1.5 If a substance, at 1.0 atm of pressure is cooled from 400 K to 200K, (see the phase diagram below) what phase change will occur? %3D 268 293 750 1000 Temperature (K) O vaporization O freezing O condensation O melting O sublimation Pressure (atm) 0.5 1.0 1.5
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
At 273 K and 1 atm pressure, one mole of an ideal gas occupies (a) Looking back at Figure 18.1, do you predict that 1 mole of an ideal gas in the middle of the stratosphere would occupy a greater or...
-
The vapour pressure of pure liquid A at 293 K is 68.8 kPa and that of pure liquid B is 82.1 kPa. These two compounds form ideal liquid and gaseous mixtures. Consider the equilibrium composition of a...
-
The phase diagram of a hypothetical substance is (a) Estimate the normal boiling point and freezing point of the substance. (b) What is the physical state of the substance under the following...
-
Find the requested numbers(s) in Problems 1116. Classify the equation as true, false, or open; and if it is open tell whether it is a conditional, identity, or contradiction. a. Three times the sum...
-
Think of your direct supervisor. Of what department is he or she a member and at what level of management is this person?
-
Willie Lohmann travels from city to city in the conduct of his business. Every other year he buys a used . car for about $12,000. The auto dealer allows about $8000 as a trade-in allowance with the...
-
Bridget applied for a TV news commentators job. She has adequate experience to satisfy the job description but was denied the job. Bridget has a substantial birthmark that covers one side of her...
-
(Financial Reporting Pressures) Presented below is abbreviated testimony from Troy Normand in the WorldCom case. He was a manager in the corporate reporting department and is one of five individuals...
-
Kericho Sports Club has been operating for some time now. The treasurer analysed the cash book and produced the following Receipts and Payments account for the year ended 31st Dec. 2018. Receipts...
-
A year ago, Rebecca purchased 100 shares of Havad stock for $25 per share. Yesterday, she placed a limit order to sell her stock at a price of $30 per share before the market opened. The stocks price...
-
4. What tax return must be filed for the estate if income is earned before properties are distributed to the beneficiaries. What option is available to minimize tax? 5. How will Lynn's registered...
-
The sprung parts of a vehicle weigh 9.79 KN, its center of gravity is 106.7 cm behind the front axle, and the wheelbase is 228.6 cm. The combined stiffness of the springs of the front suspension is...
-
Consider a small open economy (e.g. the Netherlands) producing two goods, clothing and food. The clothing industry uses capital (K) and labor (LC) as inputs, while the food industry uses land (La)...
-
There are a few points that I hope to bring into the light for you concerning psychotherapeutic drugs. In some cases, these drugs are necessary as the individual is suffering severely. In most cases,...
-
Question it is the bankruptcy question ASKED: how much money is left for unsecured debt. Also, list the debt that is unsecured one by one Peter was the child of German immigrants, and at one time had...
-
Consider the recurrence that naturally arises in some divide-and-conquer algorithms: T(1)= 1, T(n) = 4T(n/2) + n for n > 1 [a] Explain what the initial condition and the various parts of this...
-
An electric kettle consumes 5A current when connected to a voltage source of 220V. (a) Calculate the Power of the electric kettle? (b) If 1.2 kg water at 25C is heated in the electric kettle, how...
-
Prove the formula for (d/dx)(cos-1x) by the same method as for (d/dx)(sin-1x).
-
The Eyring equation can also be applied to physical processes. As an example, consider the rate of diffusion of an atom stuck to the surface of a solid. Suppose that in order to move from one site to...
-
Discuss the physical origin of quantization energy for a particle confined to moving inside a one-dimensional box or on a ring.
-
Studies of combustion reactions depend on knowing the concentrations of H atoms and HO radicals. Measurements on a flow system using EPR for the detection of radicals gave information on the...
-
You're bouncing up and down on a trampoline. After you have left the trampoline and are moving upward, your apparent weight is A. More than your true weight. B. Less than your true weight. C. Equal...
-
A mountain biker is climbing a steep 20 slope at a constant speed. The cyclist and bike have a combined weight of800 N. Referring to Figure 5.11 for guidance, what can you say about the magnitude of...
-
Rank in order, from largest to smallest, the size of the friction forces in the five different situations (one or more friction forces could be zero). The box and the floor are made of the same...

Study smarter with the SolutionInn App


