If the following molecules undergo free radical substitution, what is the order with which the hydrogen...
Fantastic news! We've Found the answer you've been seeking!
Question:
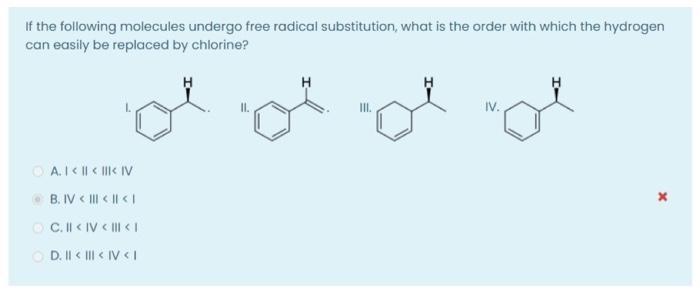
Transcribed Image Text:
If the following molecules undergo free radical substitution, what is the order with which the hydrogen can easily be replaced by chlorine? III. IV. A. | < || < ||| < IV B. IV < | < | If the following molecules undergo free radical substitution, what is the order with which the hydrogen can easily be replaced by chlorine? III. IV. A. | < || < ||| < IV B. IV < | < |
Expert Answer:
Answer rating: 100% (QA)
Option D Since freeradical substitution reaction rate depends on stability o... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
If the following molecules is treated with acid catalyst, an intra molecular esterification reaction occurs. What is the structure of the produced? (Intra molecular means within the samemolecule?
-
If one hydrogen in a hydrocarbon is replaced by a halogen atom, the number of isomers that exist for the substituted compound depends on the number of types of hydrogen in the original hydrocarbon....
-
The radical reaction of propane with chlorine yields (in addition to more highly halogenated compounds)1-chloropropane and 2-chloropropane. Cl
-
Prepare an Orientation Plan for the select position utilizing the theories discussed in the text. Discuss appropriate and effective orientation plans and your decision-making process for selecting a...
-
Contrast applied research and basic research.
-
Find the volume of the following solids using triple integrals. The region bounded by the surfaces z = e y and z = 1 over the rectangle {(x, y): 0 x 1, 0 y ln 2} In 21 X
-
Sevilla Chemicals earned $1 billion in after-tax operating income on capital invested of $5 billion last year. The firms cost of equity is 12%, its debt-to-capital ratio is 25%, and the after-tax...
-
The following monthly data are taken from Ramirez Company at July 31: Sales salaries, $300,000; Office salaries, $60,000; Federal income taxes withheld, $90,000; State income taxes withheld, $20,000;...
-
Hello! Can you please help me to understand the following problem? I have attached the blank entry sheet for reference. Thank you so much in advance! 11) Ziggy Company uses the straight-line method...
-
Bowman Specialists Inc. (BSI) manufactures specialized equipment for polishing optical lenses. There are two modelsone (A25) principally used for fine eyewear and the other (A10) for lenses used in...
-
EvTel is preparing to enter the smart home market. Thus, they are conducting market research in Istanbul. The company is planning to analyze the past 3 years' smart home device sales data with...
-
Laurence was born with Prader - Willi Syndrome. He has supportive family and friends, but he has very low self - esteem and low confidence. In extreme cases, he just locks himself in his bedroom....
-
You are creating a portfolio of two stocks. The first one has a standard deviation of 35% and the second one has a standard deviation of 30%. The correlation coefficient between the returns of the...
-
The Walters accumulated $ 2 5 6 0 0 0 during more than 4 0 years of work They originally deposited this money in a 5 - year time deposit earning 5 % and used the income for living expenses on...
-
With the maturity and fv, respectively, of a zero coupon bond you are considering that will allow you to immunize your tuition obligation of $50,000 per year at the end of the next two years. Bonds...
-
Required rate of return on the market is 1 0 % , the risk - free rate is 8 % , firm\'s beta is 3 and the firm\'s tax rate is 3 0 % . What is firm\'s the cost of common equity?
-
Organisational learning can't take place without A ) organistional privacy for employees or B ) chain of command or C ) shered vision of future or D ) stable structure or hierarchy
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Imine formation is reversible. Show all the steps involved in the acid-catalyzed reaction of an imine with water (hydrolysis) to yield an aldehyde or ketone plus primary amine.
-
The herbicide acifluorfen can be prepared by a route that begins with reaction between a phenol and an aryl fluoride. Propose a mechanism. NO2 CO2CH3 NO2 co,CH3 NO2 .CO2H DMSO HO F3C F3C CI FC...
-
The following information is available for October 2005. Beginning cash balance for October is expected to be \($60,000\). The company intends to maintain a cash balance of at least \($50,000\). The...
-
The following information is available for the Ortega Company for the first quarter of 2005: Beginning cash balance for January 2005 is expected to be \($20,000\). The company intends to maintain a...
-
The following information is available for the Perlmuter Printing Supply Company. Required: a. Prepare budgeted balance sheets for July, August, and September 2004. b. Prepare budgeted statements of...

Study smarter with the SolutionInn App


