If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas...
Fantastic news! We've Found the answer you've been seeking!
Question:


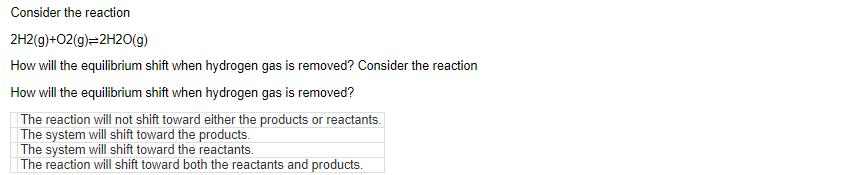

Transcribed Image Text:
If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas is added? If the reaction is at equilibrium, in what direction will the reaction shift if gas is added? The reaction will remain unchanged. The reaction will shift to produce both reactants and products. The reaction will shift to produce products. The reaction will shift to produce reactants. If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? The reaction will shift toward both reactants and products. The reaction will shift toward the reactants. The reaction will not shift toward products or reactants. The reaction will shift toward the products. Consider the reaction 2H2(g)+O2(g)=2H2O(g) How will the equilibrium shift when hydrogen gas is removed? Consider the reaction How will the equilibrium shift when hydrogen gas is removed? The reaction will not shift toward either the products or reactants. The system will shift toward the products. The system will shift toward the reactants. The reaction will shift toward both the reactants and products. Consider the exothermic reaction Fe203(s)+2Al(s) A1203(s)+2Fe(s) In what direction will the equilibrium shift when heat is removed? Consider the exothermic reaction In what direction will the equilibrium shift when heat is removed? The reaction will not shift toward either the products or reactants. The equilibrium will shift toward the reactants. The reaction will shift toward both the reactants and products. The equilibrium will shift toward the products. If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas is added? If the reaction is at equilibrium, in what direction will the reaction shift if gas is added? The reaction will remain unchanged. The reaction will shift to produce both reactants and products. The reaction will shift to produce products. The reaction will shift to produce reactants. If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? The reaction will shift toward both reactants and products. The reaction will shift toward the reactants. The reaction will not shift toward products or reactants. The reaction will shift toward the products. Consider the reaction 2H2(g)+O2(g)=2H2O(g) How will the equilibrium shift when hydrogen gas is removed? Consider the reaction How will the equilibrium shift when hydrogen gas is removed? The reaction will not shift toward either the products or reactants. The system will shift toward the products. The system will shift toward the reactants. The reaction will shift toward both the reactants and products. Consider the exothermic reaction Fe203(s)+2Al(s) A1203(s)+2Fe(s) In what direction will the equilibrium shift when heat is removed? Consider the exothermic reaction In what direction will the equilibrium shift when heat is removed? The reaction will not shift toward either the products or reactants. The equilibrium will shift toward the reactants. The reaction will shift toward both the reactants and products. The equilibrium will shift toward the products.
Expert Answer:
Answer rating: 100% (QA)
ANSWER Concept and reason The concepts used to solve this question are to determine effect of the ch... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The following reaction is at equilibrium: N2 + 3H2 + 2NH3 The Keq at a particular temperature is 13.7. If the equilibrium [N2] is 0.055 M and the equilibrium [H2] is 1.62 M, what is the equilibrium...
-
The following reaction is at equilibrium: 4HBr + O2 + 2H2O + 2Br2 If [HBr] is 0.100 M, [O2] is 0.250 M, and [H2O] is 0.0500 M at equilibrium, what is [Br2] at equilibrium if the Keq is 0.770?
-
Consider the reaction of hydrogen gas with oxygen gas: Assuming complete reaction, which of the diagrams shown next represents the amounts of reactants and products left after the reaction? 2H2(g) +...
-
Q-1 A motor car shaft consists of a steel tube 30 mm internal diameter and 4 mm thick. The engine develops 10 kW at 2000 r.p.m. Find the maximum shear stress in the tube when the power is transmitted...
-
Exhibit 6.5 describes the key financial ratios Standard & Poors analysts use to assess credit risk and assign credit ratings to industrial companies. The same financial ratios for three firms follow....
-
Mark Martinko has been a class A racquetball player for the past five years, and one of his biggest goals is to own and operate a racquetball facility. Unfortunately, Mark's thinks that the chance of...
-
Respondents Varsity Brands, Inc., Varsity Spirit Corporation, and Varsity Spirit Fashions & Supplies, Inc., design, make, and sell cheerleading uniforms. Respondents have obtained or acquired more...
-
Sara Yu, CPA, was retained by Info Cable to prepare financial statements for April 2014. Yu accumulated all the ledger balances per Info's records and found the following. Sara Yu reviewed the...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large central IS organization with several operational relational databases at its...
-
The towline exerts a force of P = 4 kN at the end of the 20-m-long crane boom. If x = 25 m, determine the position ? of the boom so that this force creates a maximum moment about point O. What is...
-
Reflect on how either curiosity has appeared in your life recently. Develop a 2-3 minute presentation that you will share with the class about your experience. You may use the resources listed below...
-
Ladder schematics is shown below. Choose which NO or NC Inputs combination will activate the Output coil. a. 11, 12, 14, 15, 16 O b. 11,17 O c. 14, 15, 16, 17 Od. 11, 12, 13, 14, 15, 16 O e. 11, 12,...
-
Create Restaurant Java Programming Using Array List: You will use cases to enter data and inside of the case 1 you should have choices using if - else conditions. If you press 1 then it will ask...
-
Your task is to make a class for a binary heap of integers, then use that class to implement heapsort. The document " Notes about Heaps " discusses the calculation-based method of storing a binary...
-
How did the expansion of scientific knowledge shape and propel the emergence and evolution of sociology as a discipline?
-
11. Use the attributes shown in Figure P3.17 and write the data dictionary using the format shown in Table P3.22. Table name: PRODUCT Primary key: PROD_CODE Foreign key: VEND_CODE PROD_CODE PROD...
-
Arvin Singh is a retired factory worker who invented a rectangular case with wheels on one end and a retractable handle on the other. The suitcase can hold about four days' worth of clothes and be...
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Which of the indicators in Fig. could be used for doing the titrations in Exercises 66 and 68? Fig Pheadl Red
-
Isocyanic acid (HNCO) can be prepared by heating sodium cyanate in the presence of solid oxalic acid according to the equation 2NaOCN(s) + H2C2O4(s) 2HNCO(l) + Na2C2O4(s)
-
Draw the Lewis structure of O2F2. Assign oxidation states and formal charges to the atoms in O2F2. The com-pound O2F2 is a vigorous and potent oxidizing and fluo-rinating agent. Are oxidation states...
-
State whether you think a business would recognise the following as an expense. When would it be recognised? a Depreciation $350 b Wages to be paid in the next month $1200 c Electricity bill due to...
-
What is the difference between operating expenses and non-operating expenses. Give examples of each.
-
What is the link between the income statement and the statement of owners equity?

Study smarter with the SolutionInn App


