If this reaction releases 489 kJ of energy, how many grams of HO are formed? Show...
Fantastic news! We've Found the answer you've been seeking!
Question:
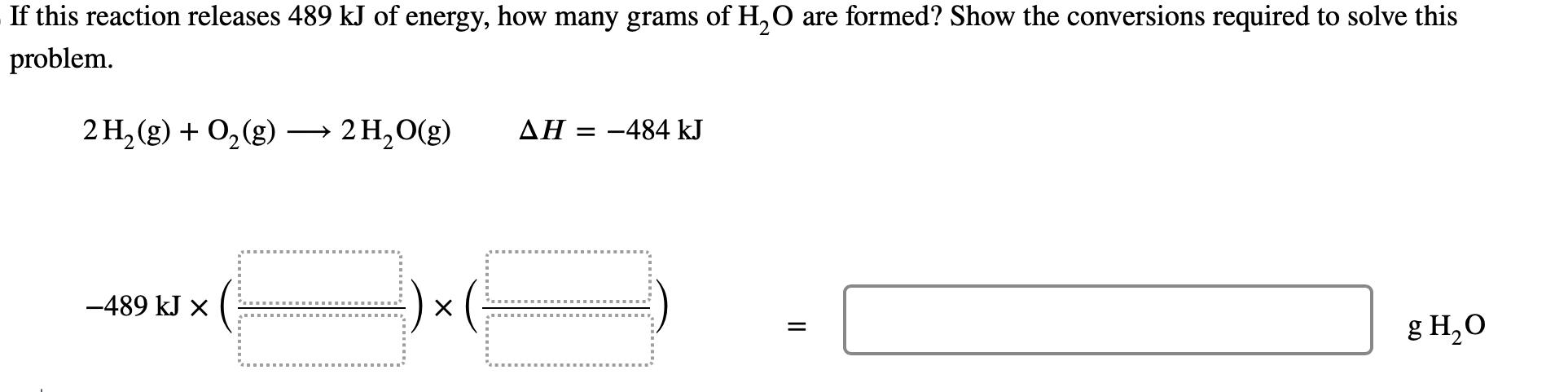
Transcribed Image Text:
If this reaction releases 489 kJ of energy, how many grams of H₂O are formed? Show the conversions required to solve this problem. 2 H₂(g) + O₂(g) → 2H₂O(g) -489 kJ X :) × ( AH = -484 kJ || g H₂O If this reaction releases 489 kJ of energy, how many grams of H₂O are formed? Show the conversions required to solve this problem. 2 H₂(g) + O₂(g) → 2H₂O(g) -489 kJ X :) × ( AH = -484 kJ || g H₂O
Expert Answer:
Answer rating: 100% (QA)
d L Solution Given Reaction 241g Og 21org AH 454 KJ from this reacti... View the full answer

Related Book For 

Posted Date:
Students also viewed these economics questions
-
How many grams of N2 will react if 100.0 kJ of energy are supplied? N2(g) + O2(g) ( 2NO(g)H = 180.6 kJ
-
How many grams of sodium succinate (formula weight 140 g mol-1) and disodium succinate (formula weight 162 g mol-1) must be added to 1 L of water to produce a solution with pH 6.0 and a total solute...
-
Show that if G and H are isomorphic directed graphs, then the converses of G and H (defined in the preamble of Exercise 67 of Section 10.2) are also isomorphic.
-
Please answer the following. Show your calculations for potential partial credit. Assume the expected return on the market is 14 percent and the risk-free rate is 4 percent. A.)What is the expected...
-
On June 30, 2012, your client, Ferry Company, was granted two patents covering plastic cartons that it had been producing and marketing profitably for the past 3 years. One patent covers the...
-
Amal, Bimal and Tamal are partners in a firm sharing profits and losses as Amal 60%, Bimal 30% and Tamal 10%. They agreed to dissolve the firm when the Balance Sheet was as under: Land and Building...
-
Match threats in the first column to appropriate control procedures in the second column. Threat 1. Sending incorrect merchandise Control Procedure a. Perform a completeness check 2. Crediting...
-
Kitchen Kings Toledo plant manufactures three product lines, all multi-burner, ceramic cook tops. The plants three product models are the Regular (REG). the Advanced (ADV), and the Gourmet (GMT)....
-
5 1 4 + x 2.7 0.6
-
For the liquid storage system shown in Fig. E, it is desired to control liquid levels h1 and h2 by adjusting volumetric flow rates q1 and q2. Flow rate q6 is the major disturbance variable. The...
-
The company Quicksilver has the following abbreviated Income Statement and Balance Sheet: Net sales 1.000.000 COGS 500.000 Operating Expenses 20.000 Income tax 72.000 Net Income 408.000 BALANCE SHEET...
-
A graduating senior with a major in accounting is considering the following job offers. a. From a "Big Six" CPA firm. b. From a small local CPA firm. c. From a large corporation with training to...
-
The understanding of internal control that relates to a financial statement assertion should be used to do all of the following except a. Determine inherent risk for that assertion. b. Identify types...
-
Let $S^{2}$ denote a sphere of unit radius, centered at the origin of $\mathbb{R}^{3}$. What is the group of transformations that leave it invariant? Next, let $S_{(1)}^{2}$ be a sphere of unit...
-
Which of the following internal control procedures would an entity most likely use to assist in satisfying the completeness assertion related to long-term investments? a. Senior management verifies...
-
Define the following terms: a. Ethics. b. Ethical commitment. c. Ethical awareness. d. Ethical competency.
-
1. When and why has the concept of sustainable development evolved? 2. What is the definition of sustainable development? 3. What are the key features of sustainable development? 4. What elements...
-
Make an argument that Williams had a right to delay the closing until after August 1.
-
Solid phosphorus pentabromide, PBr5, has been shown to have the ionic structure [PBr4+][Br]. Write the electron-dot formula of the PBr4+ cation.
-
Describe in words how to obtain the formula mass of a compound from the formula.
-
Seawater contains 0.0065% (by mass) of bromine. How many grams of bromine are there in 2.50 L of seawater? The density of seawater is 1.025 g/cm3.
-
Decade, Inc., recorded certain capital stock transactions shown in the following journal entries: (1) issued common stock for \(\$ 25\) cash per share, (2) purchased treasury shares at \(\$ 40\) per...
-
Label each of the following characteristics of a corporation as either an (A) advantage or a (D) disadvantage: a. Limited liability b. Taxation c. Regulations d. Transferability of ownership
-
The following information relates to Ontario Components, Inc.: a. Calculate the company's return on common stockholders' equity for 2018 and 2019. b. Calculate the company's dividend yield for 2018...

Study smarter with the SolutionInn App


